QUESTION IMAGE
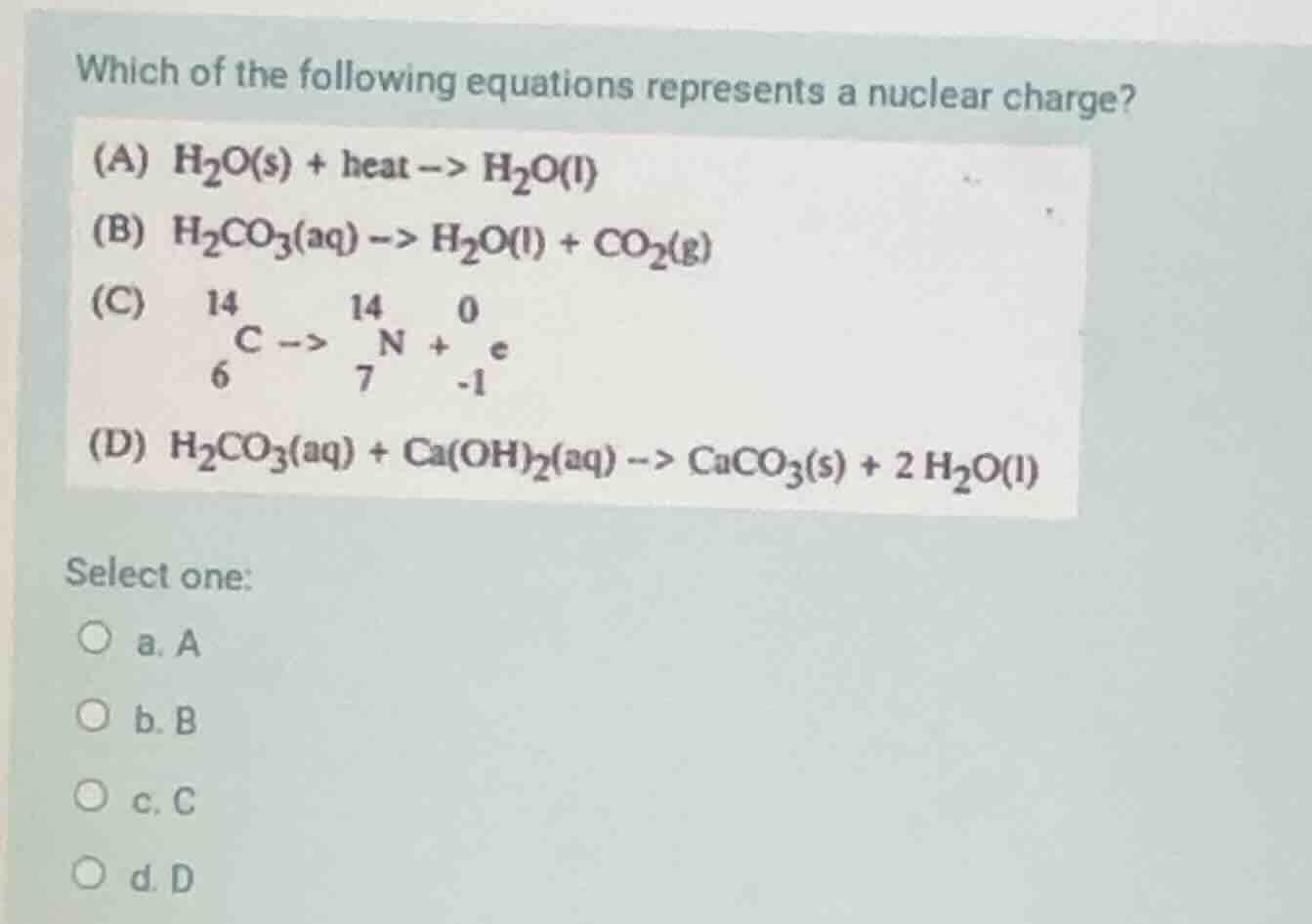
Question
which of the following equations represents a nuclear charge? (a) $\ce{h_{2}o(s) + heat -> h_{2}o(l)}$ (b) $\ce{h_{2}co_{3}(aq) -> h_{2}o(l) + co_{2}(g)}$ (c) $\ce{_{6}^{14}c -> _{7}^{14}n + _{-1}^{0}e}$ (d) $\ce{h_{2}co_{3}(aq) + ca(oh)_{2}(aq) -> caco_{3}(s) + 2h_{2}o(l)}$ select one: a. a b. b c. c d. d
A nuclear charge reaction involves changes in the nucleus of an atom, altering the atomic number or mass number. Option A is a phase change, B is a chemical decomposition, D is an acid-base reaction—all are chemical changes with no nuclear alterations. Option C shows carbon-14 decaying into nitrogen-14, with a change in atomic number (from 6 to 7) and emission of a beta particle, which is a nuclear reaction involving nuclear charge change.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
c. C