QUESTION IMAGE
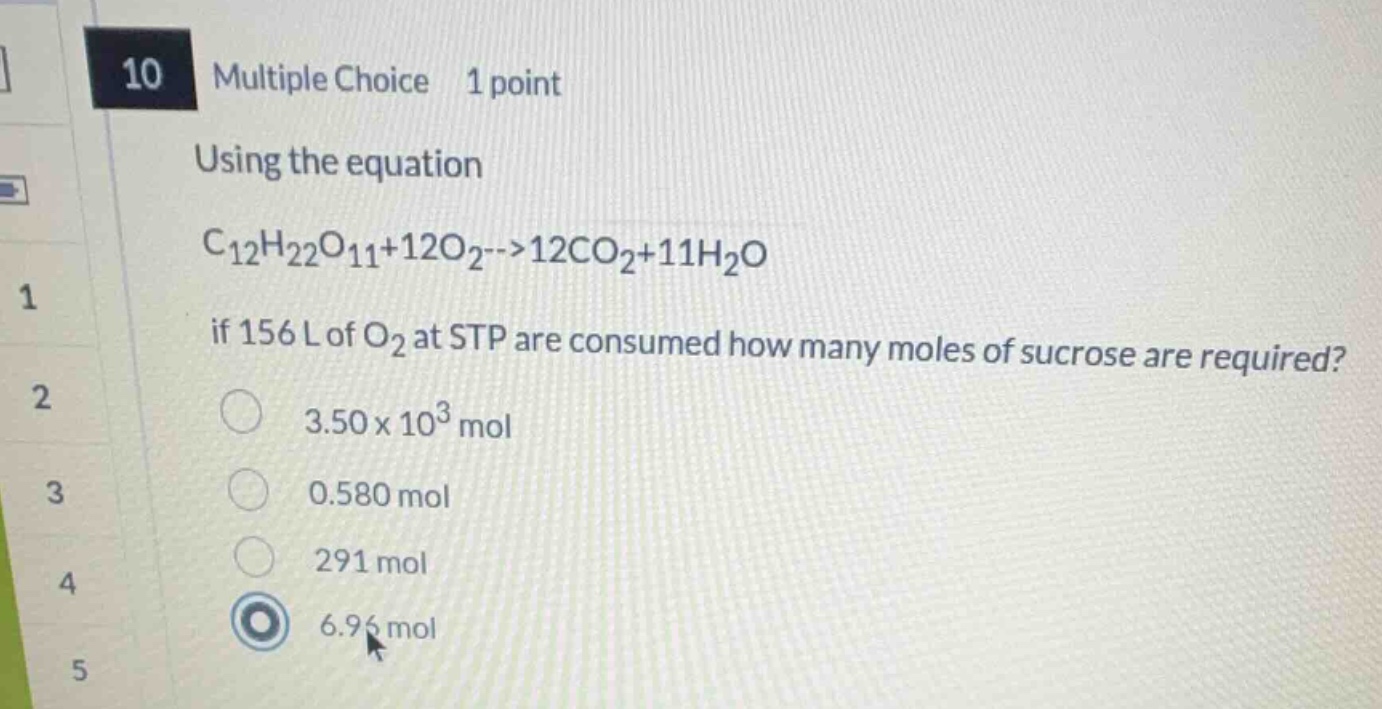
Question
10 multiple choice 1 point
using the equation
$ce{c_{12}h_{22}o_{11} + 12o_{2} -> 12co_{2} + 11h_{2}o}$
if 156 l of $ce{o_{2}}$ at stp are consumed how many moles of sucrose are required?
$3.50 \times 10^{3}$ mol
0.580 mol
291 mol
6.96 mol
Step1: Find moles of $\text{O}_2$
At STP, 1 mol gas = 22.4 L. Moles of $\text{O}_2$: $\frac{156}{22.4}$
Step2: Mole ratio of sucrose to $\text{O}_2$
From reaction: $\frac{n(\text{C}_{12}\text{H}_{22}\text{O}_{11})}{n(\text{O}_2)}=\frac{1}{12}$
Step3: Calculate moles of sucrose
$n(\text{C}_{12}\text{H}_{22}\text{O}_{11})=\frac{156}{22.4\times12}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.580 mol