QUESTION IMAGE
Question
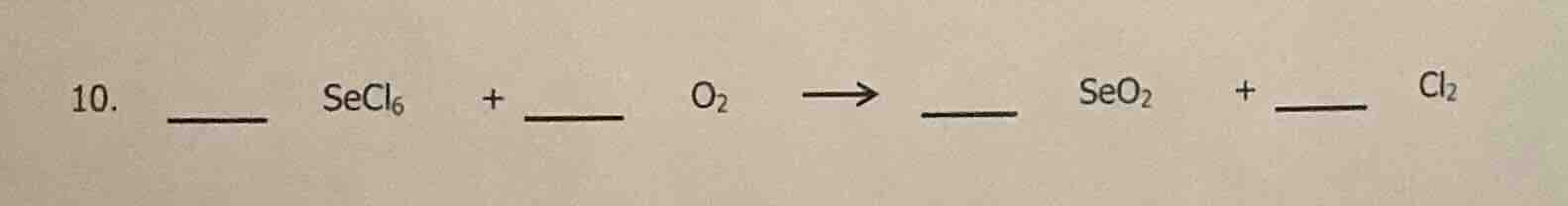
- __ secl₆ + o₂ → seo₂ + __ cl₂
Step1: Balance Se
There is 1 Se in \( \text{SeCl}_6 \) and 1 Se in \( \text{SeO}_2 \), so the coefficient for \( \text{SeCl}_6 \) and \( \text{SeO}_2 \) related to Se is 1 for now. Let's set the coefficient of \( \text{SeCl}_6 \) as \( x = 1 \), then the coefficient of \( \text{SeO}_2 \) should also be 1 to balance Se.
Step2: Balance Cl
In \( \text{SeCl}_6 \), there are 6 Cl atoms. In \( \text{Cl}_2 \), each molecule has 2 Cl atoms. So the number of \( \text{Cl}_2 \) molecules needed is \( \frac{6}{2}=3 \). So the coefficient of \( \text{Cl}_2 \) is 3.
Step3: Balance O
In \( \text{SeO}_2 \), there are 2 O atoms. In \( \text{O}_2 \), each molecule has 2 O atoms. So the number of \( \text{O}_2 \) molecules needed is \( \frac{2}{2}=1 \). So the coefficient of \( \text{O}_2 \) is 1.
Now we check the equation: \( 1\text{SeCl}_6 + 1\text{O}_2
ightarrow 1\text{SeO}_2 + 3\text{Cl}_2 \)
- Se: 1 on left, 1 on right.
- Cl: 6 on left (\( 1\times6 \)), 6 on right (\( 3\times2 \)).
- O: 2 on left (\( 1\times2 \)), 2 on right (\( 1\times2 \)).
It is balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1, 1, 1, 3 (The coefficients for \( \text{SeCl}_6 \), \( \text{O}_2 \), \( \text{SeO}_2 \), \( \text{Cl}_2 \) respectively)