QUESTION IMAGE
Question
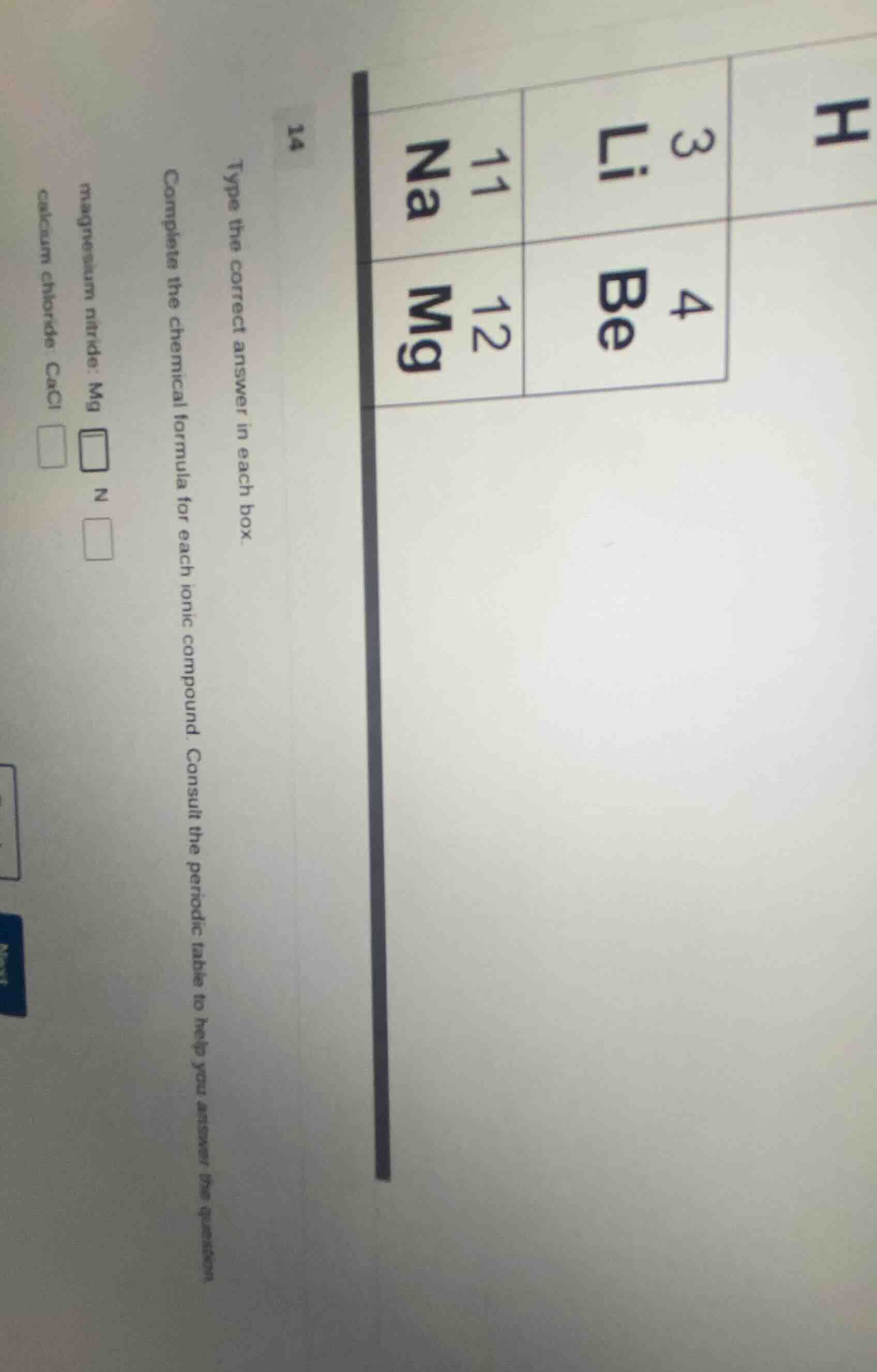
14
type the correct answer in each box.
complete the chemical formula for each ionic compound. consult the periodic table to help you answer the question
magnesium nitride: mg n
calcium chloride: cacl
Magnesium Nitride:
Step1: Determine ion charges
Magnesium (Mg) is in group 2, so it forms $Mg^{2+}$ ions. Nitrogen (N) is in group 15, so it forms $N^{3-}$ ions (since it gains 3 electrons to achieve octet).
Step2: Balance charges
To balance the charges, we need 3 $Mg^{2+}$ ions (total positive charge: $3\times2 = +6$) and 2 $N^{3-}$ ions (total negative charge: $2\times(-3) = -6$). So the formula is $Mg_3N_2$.
Calcium Chloride:
Step1: Determine ion charges
Calcium (Ca) is in group 2, so it forms $Ca^{2+}$ ions. Chlorine (Cl) is in group 17, so it forms $Cl^-$ ions (gains 1 electron).
Step2: Balance charges
To balance the $2+$ charge of Ca, we need 2 $Cl^-$ ions. So the formula is $CaCl_2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Magnesium Nitride: $\boldsymbol{Mg_3N_2}$
- Calcium Chloride: $\boldsymbol{CaCl_2}$