QUESTION IMAGE
Question
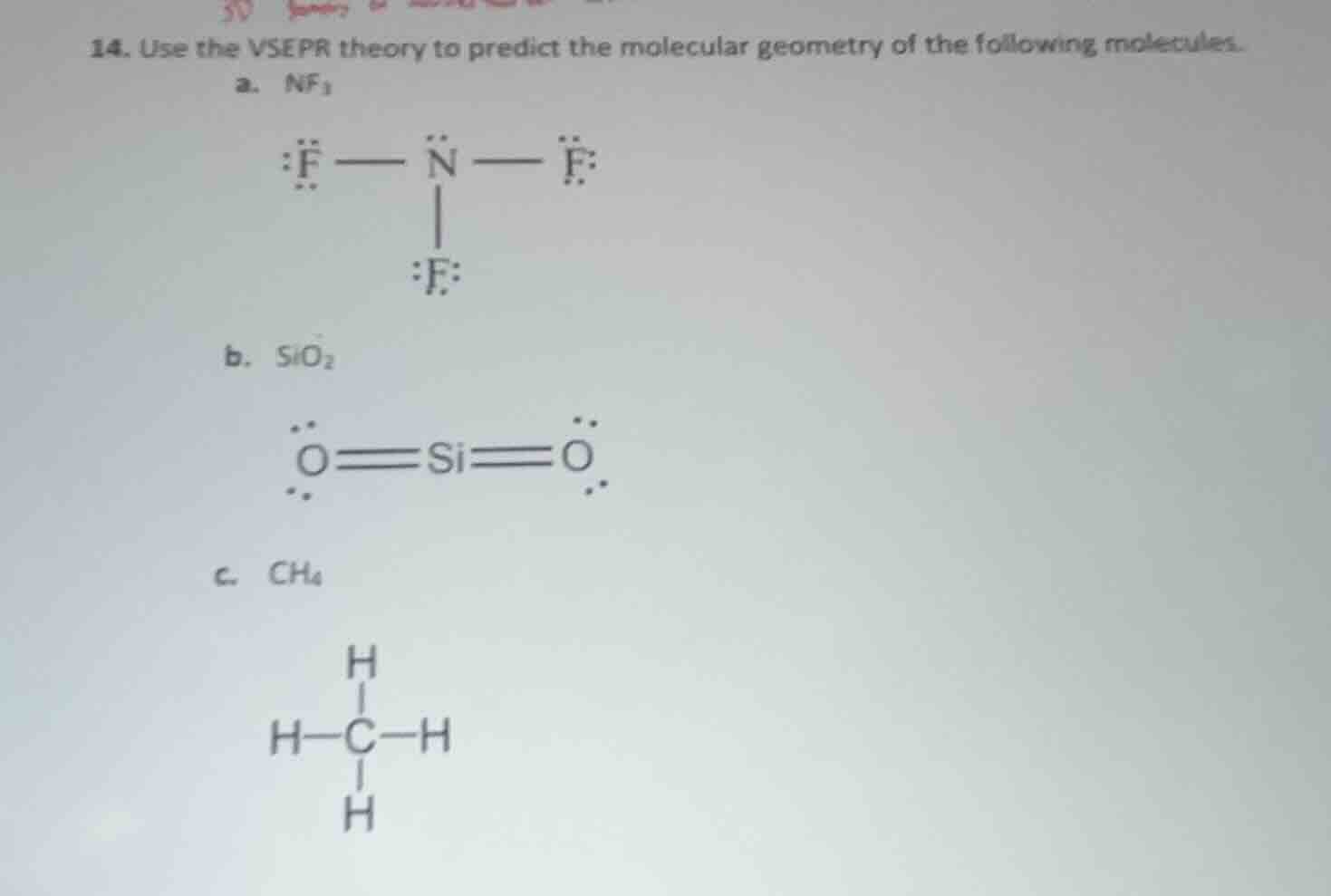
- use the vsepr theory to predict the molecular geometry of the following molecules.
a. nf₃
b. sio₂
c. ch₄
Step1: Count electron domains for NF₃
N has 3 bonding pairs + 1 lone pair = 4 electron domains.
Step2: Assign electron geometry for NF₃
4 electron domains = tetrahedral electron geometry; 1 lone pair modifies molecular geometry to trigonal pyramidal.
Step3: Count electron domains for SiO₂
Si has 2 double bonding pairs + 0 lone pairs = 2 electron domains.
Step4: Assign geometry for SiO₂
2 electron domains = linear molecular geometry (no lone pairs to modify).
Step5: Count electron domains for CH₄
C has 4 bonding pairs + 0 lone pairs = 4 electron domains.
Step6: Assign geometry for CH₄
4 electron domains, no lone pairs = tetrahedral molecular geometry.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. NF₃: Trigonal pyramidal
b. SiO₂: Linear
c. CH₄: Tetrahedral