QUESTION IMAGE
Question
- complete the ground state orbital - filling diagram and write the corresponding electron configurations below.
extension questions
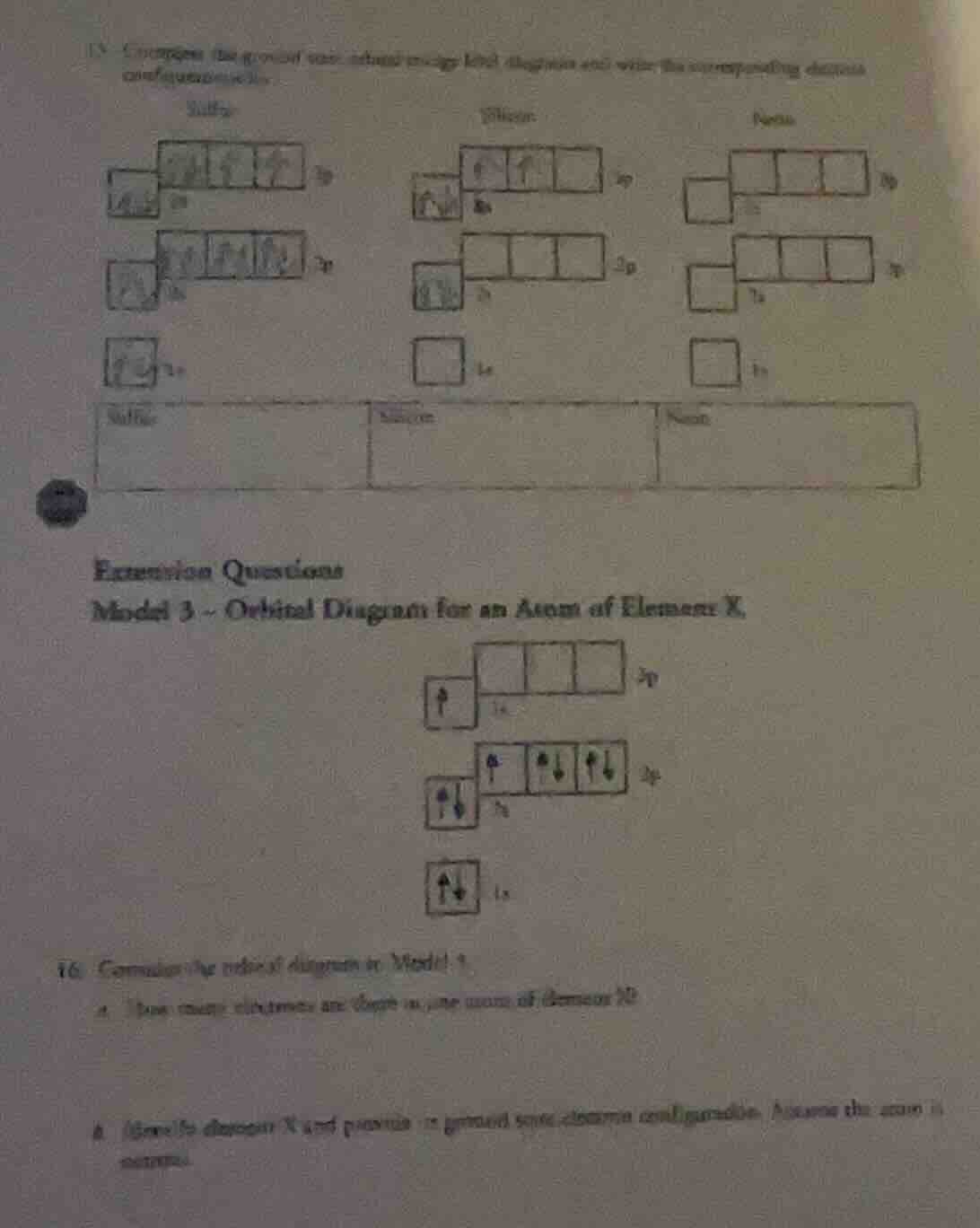
model 3 - orbital diagram for an atom of element x
- consider the orbital diagram in model 3.
a. how many electrons are there in one atom of element x?
b. identify element x and provide its ground - state electron configuration. assume the atom is neutral.
Part a: Number of electrons in element X
Step 1: Analyze 1s orbital
The 1s orbital has 2 electrons (paired, $\uparrow\downarrow$).
Step 2: Analyze 2s orbital
The 2s orbital has 2 electrons (paired, $\uparrow\downarrow$).
Step 3: Analyze 2p orbitals
- First 2p orbital: 1 electron ($\uparrow$)
- Second 2p orbital: 2 electrons (paired, $\uparrow\downarrow$)
- Third 2p orbital: 2 electrons (paired, $\uparrow\downarrow$)
Total 2p electrons: $1 + 2 + 2 = 5$
Step 4: Sum all electrons
Total electrons = 1s + 2s + 2p = $2 + 2 + 5 = 9$
Part b: Identify element X and its electron configuration
Step 1: Determine atomic number
The number of electrons (9) equals the atomic number, so element X is fluorine (F).
Step 2: Write electron configuration
Using orbital filling: 1s² (2 electrons), 2s² (2 electrons), 2p⁵ (5 electrons).
Thus, electron configuration: $1s^2 2s^2 2p^5$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
a. $\boldsymbol{9}$ electrons.
b. Element X is $\boldsymbol{\text{Fluorine (F)}}$, electron configuration: $\boldsymbol{1s^2 2s^2 2p^5}$