QUESTION IMAGE
Question
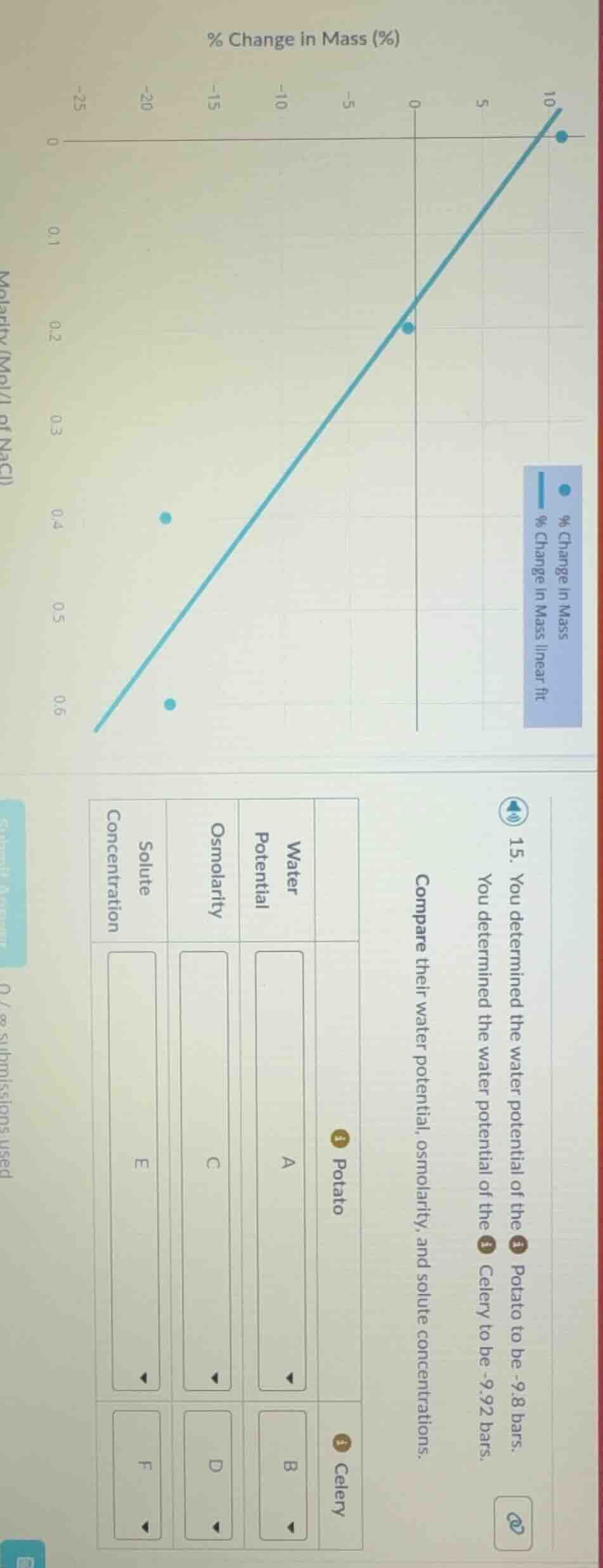
- you determined the water potential of the potato to be -9.8 bars. you determined the water potential of the celery to be -9.92 bars. compare their water potential, osmolarity, and solute concentrations. potato water potential a osmolarity c solute concentration e celery water potential b osmolarity d solute concentration f
Brief Explanations
- Water Potential: Water potential is measured directly here; the given value for Potato is -9.8 bars, and for Celery is -9.92 bars.
- Osmolarity: The osmolarity of a solution is equal to the solute concentration for ionic compounds like NaCl (since NaCl dissociates into 2 ions, but the molarity on the graph corresponds to osmolarity for this context). The linear fit crosses 0% mass change (isotonic point, where water potential matches the tissue) at ~0.18 M NaCl, so osmolarity is ~0.18 osmol/L. Since Potato and Celery have very close water potentials, their osmolarities are nearly equal, with Celery's slightly higher to match its lower water potential.
- Solute Concentration: For NaCl, solute concentration (molarity) is half the osmolarity (due to dissociation into Na⁺ and Cl⁻). For Potato, $\frac{0.18}{2}=0.09$ M; for Celery, a slightly higher ~0.095 M to align with -9.92 bars.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Potato | Celery | |
|---|---|---|
| Osmolarity | C. ~0.18 osmol/L | D. ~0.19 osmol/L |
| Solute Concentration | E. ~0.09 M NaCl | F. ~0.095 M NaCl |