QUESTION IMAGE
Question
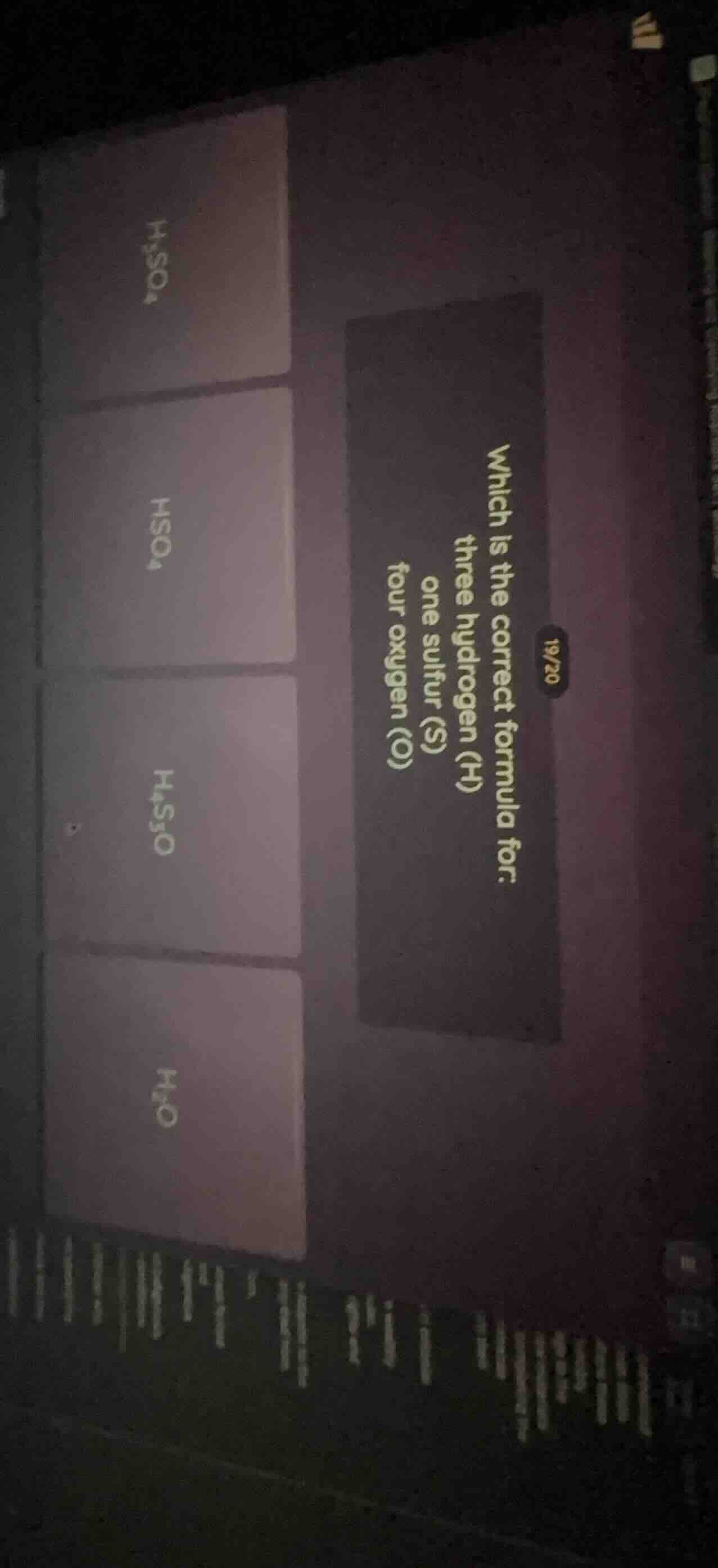
19/20 which is the correct formula for: three hydrogen (h) one sulfur (s) four oxygen (o) options: h₂so₄, hso₄, h₄s₃o, h₃o
Chemical formulas use subscripts after each element symbol to denote the number of atoms of that element. For the given counts (3 H, 1 S, 4 O), the formula should have $\text{H}_3$ for three hydrogen atoms, $\text{S}$ (no subscript means 1 atom) for sulfur, and $\text{O}_4$ for four oxygen atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
None of the provided options match the correct formula $\text{H}_3\text{SO}_4$. The options listed are $\text{H}_2\text{SO}_4$, $\text{HSO}_4$, $\text{H}_4\text{S}_3\text{O}$, and $\text{H}_2\text{O}$, none of which correspond to 3 hydrogen, 1 sulfur, and 4 oxygen atoms.