QUESTION IMAGE
Question
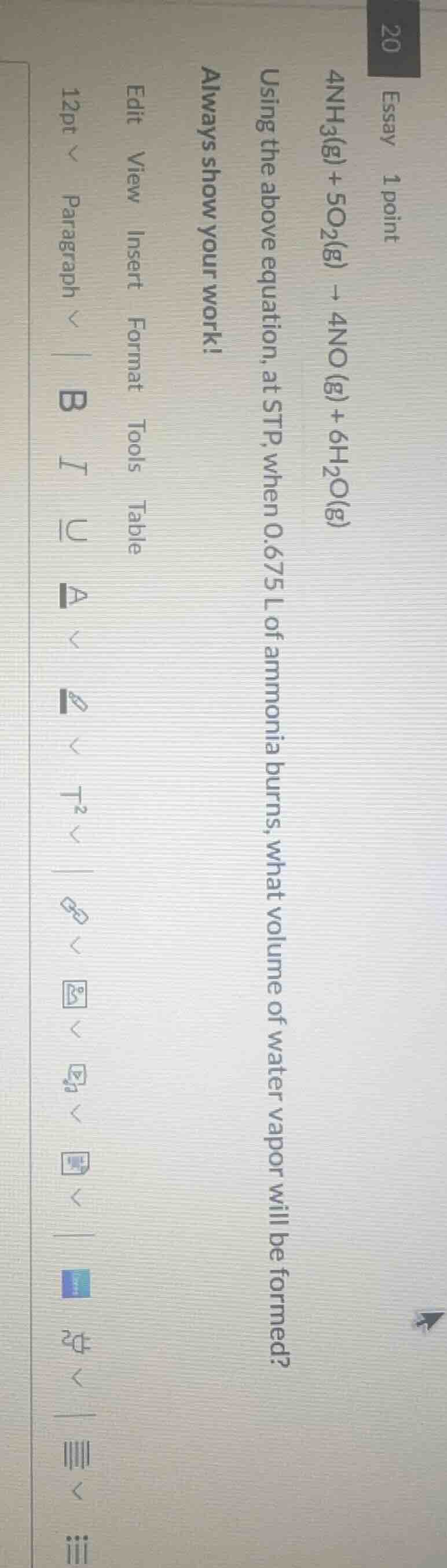
20 essay 1 point
$4nh_3(g) + 5o_2(g) \
ightarrow 4no(g) + 6h_2o(g)$
using the above equation, at stp, when 0.675 l of ammonia burns, what volume of water vapor will be formed?
always show your work!
Step1: Identify mole ratio
From the balanced equation: $\frac{V_{\ce{H2O}}}{V_{\ce{NH3}}} = \frac{6}{4}$
Step2: Calculate water vapor volume
Substitute $V_{\ce{NH3}}=0.675\ \text{L}$:
$V_{\ce{H2O}} = 0.675\ \text{L} \times \frac{6}{4}$
Step3: Compute final value
$V_{\ce{H2O}} = 0.675 \times 1.5 = 1.0125\ \text{L}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$1.01\ \text{L}$ (or $1.0125\ \text{L}$)