QUESTION IMAGE
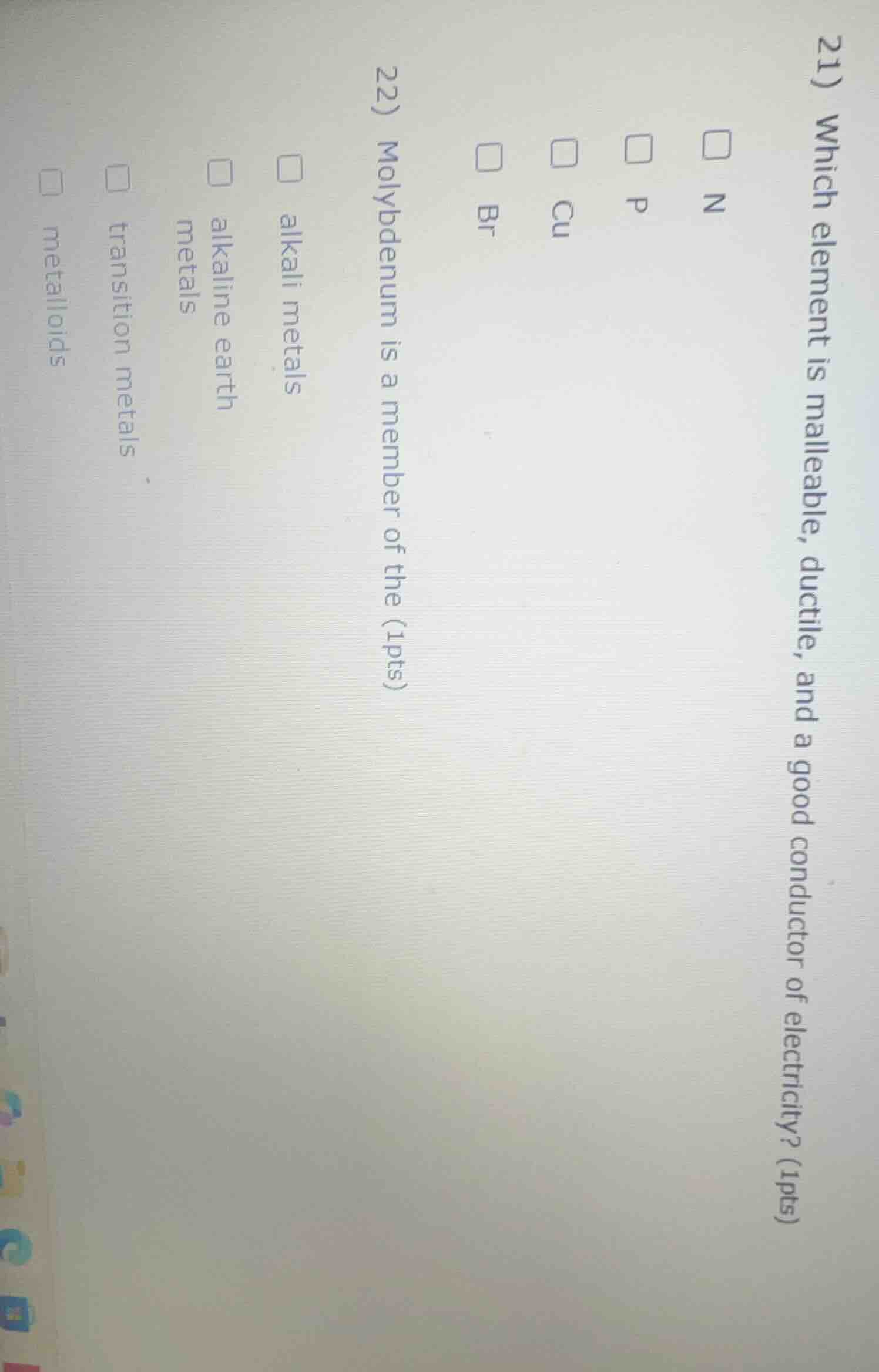
Question
- which element is malleable, ductile, and a good conductor of electricity? (1pts)
□ n
□ p
□ cu
□ br
- molybdenum is a member of the (1pts)
□ alkali metals
□ alkaline earth metals
□ transition metals
□ metalloids
For question 21: Malleability, ductility, and high electrical conductivity are key properties of metals. Nitrogen (N) and Phosphorus (P) are nonmetals, Bromine (Br) is a nonmetal liquid, while Copper (Cu) is a metal with all these properties.
For question 22: Molybdenum (Mo) has an electron configuration that places it in the d-block of the periodic table, which defines transition metals. Alkali metals are group 1, alkaline earth metals are group 2, and metalloids have properties between metals and nonmetals, which does not apply to molybdenum.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Cu
- transition metals