QUESTION IMAGE
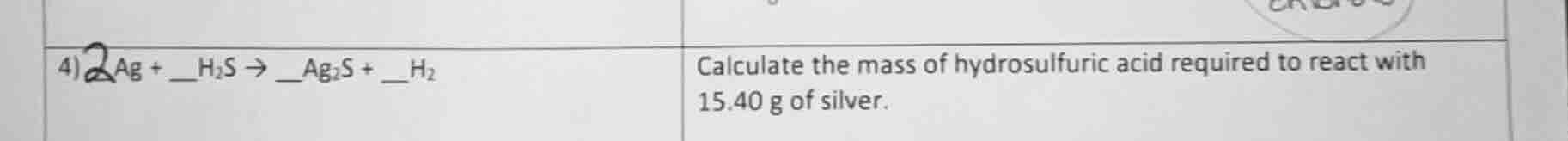
Question
- 2ag + h₂s → ag₂s + __h₂ calculate the mass of hydrosulfuric acid required to react with 15.40 g of silver.
Step1: Balance the chemical equation
First, we balance the given chemical equation: \(2\text{Ag} + \text{H}_2\text{S}
ightarrow \text{Ag}_2\text{S} + \text{H}_2\) (the coefficients are already correct as 2 for Ag, 1 for \(\text{H}_2\text{S}\), 1 for \(\text{Ag}_2\text{S}\), and 1 for \(\text{H}_2\)).
Step2: Calculate moles of Ag
The molar mass of Ag is \(107.87\space g/mol\). Given mass of Ag is \(15.40\space g\). Moles of Ag, \(n_{\text{Ag}}=\frac{\text{mass}}{\text{molar mass}}=\frac{15.40\space g}{107.87\space g/mol}\approx0.1427\space mol\).
Step3: Determine mole ratio of \(\text{H}_2\text{S}\) to Ag
From the balanced equation, the mole ratio of \(\text{H}_2\text{S}\) to Ag is \(1:2\) (1 mole of \(\text{H}_2\text{S}\) reacts with 2 moles of Ag).
Step4: Calculate moles of \(\text{H}_2\text{S}\)
Using the mole ratio, moles of \(\text{H}_2\text{S}\), \(n_{\text{H}_2\text{S}}=\frac{1}{2}\times n_{\text{Ag}}=\frac{1}{2}\times0.1427\space mol = 0.07135\space mol\).
Step5: Calculate mass of \(\text{H}_2\text{S}\)
The molar mass of \(\text{H}_2\text{S}\) is \(2\times1 + 32.07=34.07\space g/mol\). Mass of \(\text{H}_2\text{S}\), \(m = n\times\text{molar mass}=0.07135\space mol\times34.07\space g/mol\approx2.431\space g\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The mass of hydrosulfuric acid (\(\text{H}_2\text{S}\)) required is approximately \(2.43\space g\) (or more precisely \(2.431\space g\)).