QUESTION IMAGE
Question
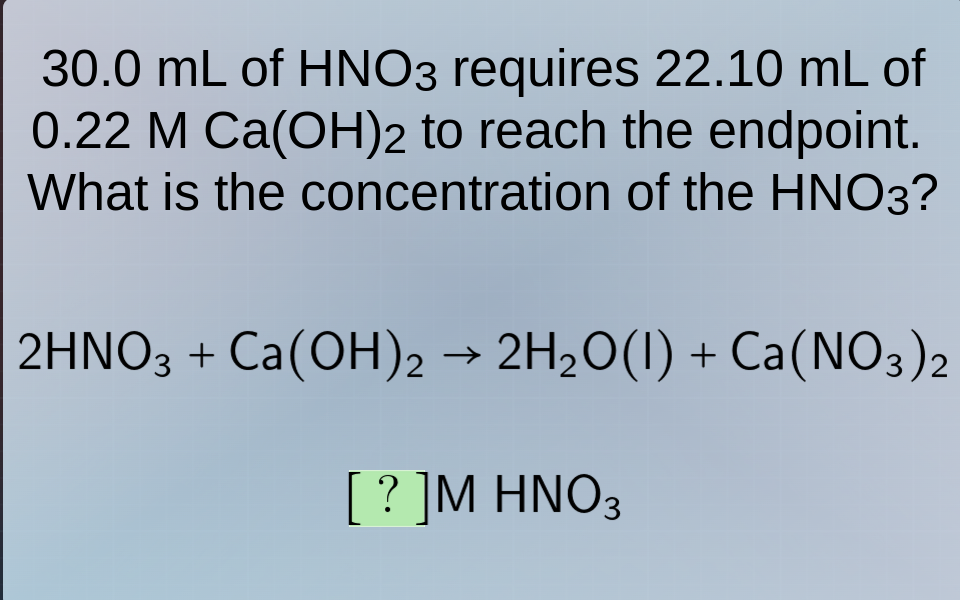
30.0 ml of hno₃ requires 22.10 ml of 0.22 m ca(oh)₂ to reach the endpoint. what is the concentration of the hno₃? 2hno₃ + ca(oh)₂ → 2h₂o(l) + ca(no₃)₂ ?m hno₃
Step1: Recall the titration formula
For acid - base titrations, the formula based on the stoichiometry of the reaction is \(n_1M_1V_1=n_2M_2V_2\), where \(n_1\) and \(n_2\) are the number of moles of acid and base (from the balanced equation), \(M_1\) and \(M_2\) are their molarities, and \(V_1\) and \(V_2\) are their volumes. From the balanced equation \(2HNO_3+Ca(OH)_2
ightarrow2H_2O + Ca(NO_3)_2\), \(n_1 = 2\) (moles of \(HNO_3\)), \(n_2=1\) (moles of \(Ca(OH)_2\)), \(V_1 = 30.0\space mL\), \(M_2=0.22\space M\), \(V_2 = 22.10\space mL\). We need to find \(M_1\) (molarity of \(HNO_3\)).
Step2: Rearrange the formula to solve for \(M_1\)
From \(n_1M_1V_1=n_2M_2V_2\), we can re - arrange it as \(M_1=\frac{n_2M_2V_2}{n_1V_1}\)
Step3: Substitute the values into the formula
Substitute \(n_1 = 2\), \(n_2 = 1\), \(M_2=0.22\space M\), \(V_2 = 22.10\space mL\), \(V_1=30.0\space mL\) into the formula:
\(M_1=\frac{1\times0.22\space M\times22.10\space mL}{2\times30.0\space mL}\)
First, calculate the numerator: \(1\times0.22\times22.10 = 0.22\times22.10=4.862\)
Then, calculate the denominator: \(2\times30.0 = 60.0\)
Now, divide the numerator by the denominator: \(M_1=\frac{4.862}{60.0}\approx0.081\space M\) (Wait, no, let's recalculate: \(0.22\times22.10 = 4.862\), \(4.862\div(2\times30.0)=4.862\div60.0\approx0.081\)? Wait, no, I made a mistake. Wait, \(n_1 = 2\) (acid), \(n_2 = 1\) (base). Wait, the formula is based on the reaction stoichiometry. The correct relation is moles of acid \(=\) moles of base \(\times\frac{n_1}{n_2}\) from the balanced equation. Moles of \(Ca(OH)_2\) \(=M_2V_2\) (in liters, but since we are using volumes in mL, the units will cancel out as long as we are consistent). Moles of \(Ca(OH)_2\) \(=0.22\space mol/L\times\frac{22.10}{1000}\space L = 0.22\times22.10\times10^{- 3}\space mol\). From the equation, 1 mole of \(Ca(OH)_2\) reacts with 2 moles of \(HNO_3\), so moles of \(HNO_3\) \(=2\times\) moles of \(Ca(OH)_2\). Then molarity of \(HNO_3\) \(=\frac{\text{moles of }HNO_3}{V_1(\text{in liters})}\)
Moles of \(Ca(OH)_2\): \(n_{Ca(OH)_2}=M_2V_2 = 0.22\space M\times0.02210\space L=0.22\times0.02210\space mol = 0.004862\space mol\)
Moles of \(HNO_3\): \(n_{HNO_3}=2\times n_{Ca(OH)_2}=2\times0.004862 = 0.009724\space mol\)
Volume of \(HNO_3\) in liters: \(V_{HNO_3}=0.0300\space L\)
Molarity of \(HNO_3\): \(M_{HNO_3}=\frac{n_{HNO_3}}{V_{HNO_3}}=\frac{0.009724\space mol}{0.0300\space L}\approx0.324\space M\)
Wait, let's do it using the first formula again. \(n_1M_1V_1=n_2M_2V_2\)
\(2\times M_1\times30.0=1\times0.22\times22.10\)
\(M_1=\frac{0.22\times22.10}{2\times30.0}=\frac{4.862}{60.0}\approx0.081\)? No, that's wrong. Wait, no, the volume units: when we use \(V\) in mL, the formula still holds because the conversion factor from mL to L is the same for both. Let's check the units: \(M=\frac{mol}{L}\), \(V\) in mL: \(n = M\times V(mL)\times10^{- 3}\). So for acid: \(n_1 = M_1\times V_1\times10^{-3}\), for base: \(n_2=M_2\times V_2\times10^{-3}\). From the reaction, \(n_1 = 2n_2\) (since 2 moles of acid react with 1 mole of base). So \(M_1\times V_1\times10^{-3}=2\times(M_2\times V_2\times10^{-3})\). The \(10^{-3}\) cancels out, so \(M_1V_1 = 2M_2V_2\), so \(M_1=\frac{2M_2V_2}{V_1}\)
Ah! I had the stoichiometry wrong in the first formula rearrangement. From the balanced equation, 2 moles of \(HNO_3\) react with 1 mole of \(Ca(OH)_2\), so moles of \(HNO_3 = 2\times\) moles of \(Ca(OH)_2\). So \(M_1V_1=2M_2V_2\) (since \(n = MV\), and \(n_{HNO_3}=2n_{Ca(OH)_2}\))
So \(M_1=\frac{2M_2V_2}{V_1}\)
Now subst…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(0.32\space M\) (or more precisely \(0.324\space M\))