QUESTION IMAGE
Question
2/50
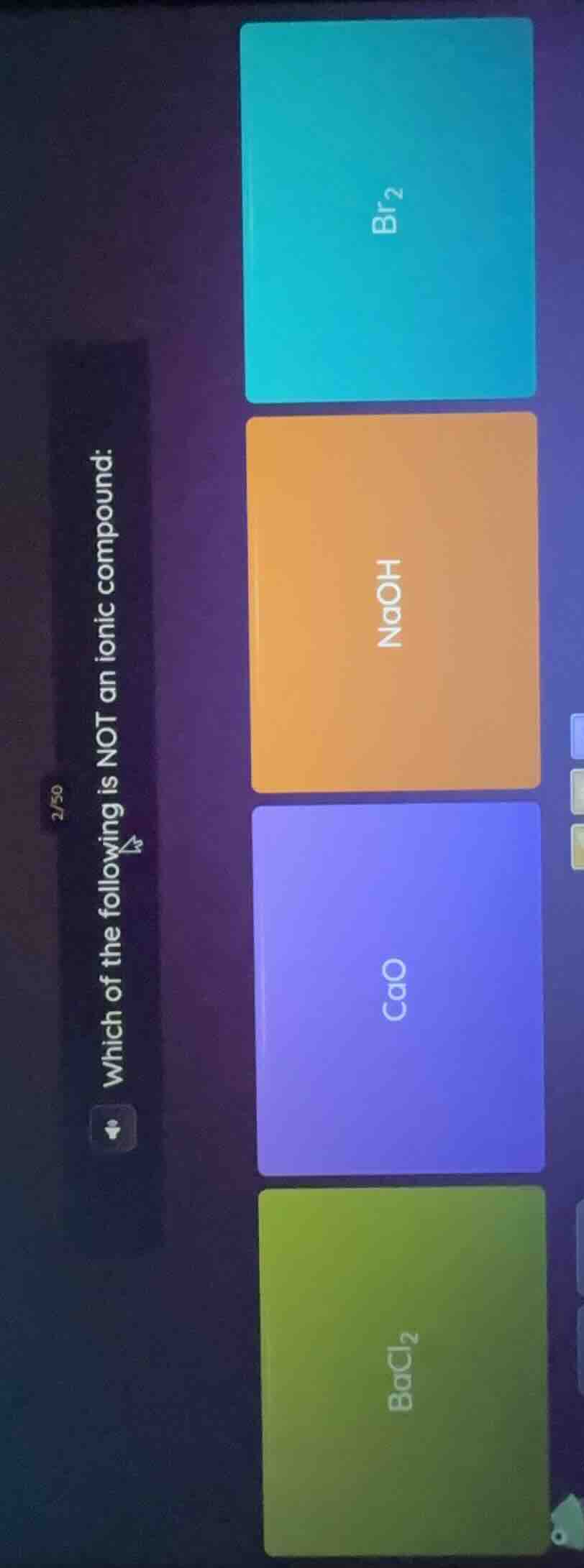
which of the following is not an ionic compound:
br₂
naoh
cao
bacl₂
Brief Explanations
Ionic compounds form between metals and nonmetals (or polyatomic ions) via electrostatic attraction of charged ions.
- $\text{BaCl}_2$: Barium (metal) + chlorine (nonmetal) = ionic.
- $\text{CaO}$: Calcium (metal) + oxygen (nonmetal) = ionic.
- $\text{NaOH}$: Sodium (metal) + hydroxide (polyatomic ion) = ionic.
- $\text{Br}_2$: Two bromine (nonmetal) atoms form a covalent molecular substance, no ions present.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{Br}_2$