QUESTION IMAGE
Question
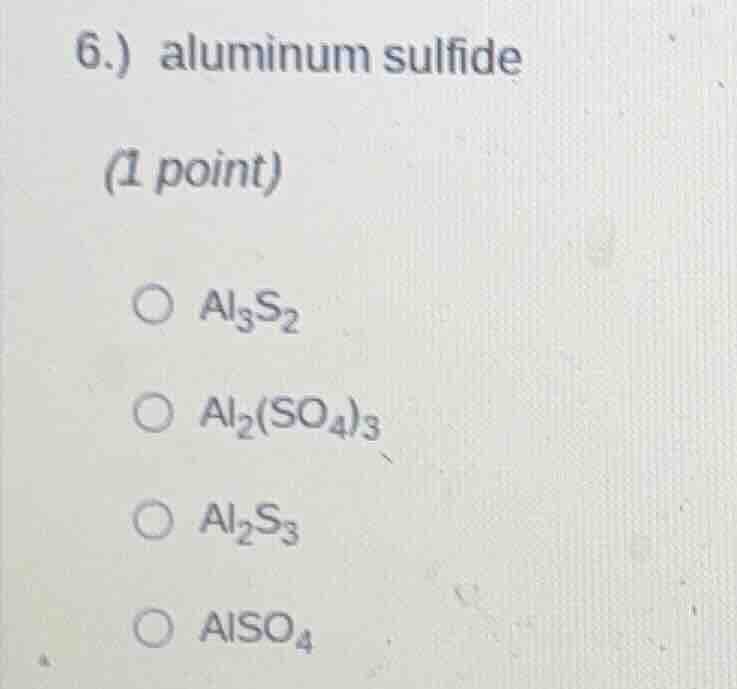
6.) aluminum sulfide
(1 point)
○ $al_3s_2$
○ $al_2(so_4)_3$
○ $al_2s_3$
○ $also_4$
Step1: Identify ion charges
Aluminum forms $\text{Al}^{3+}$; sulfide is $\text{S}^{2-}$.
Step2: Balance charge
Find least common multiple of 3 and 2: 6. Need 2 $\text{Al}^{3+}$ ($2 \times 3+ = 6+$) and 3 $\text{S}^{2-}$ ($3 \times 2- = 6-$) to balance.
Step3: Write formula
Combine ions in balanced ratio: $\text{Al}_2\text{S}_3$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $\text{Al}_2\text{S}_3$