QUESTION IMAGE
Question
apply: balancing equations
google classroom microsoft teams
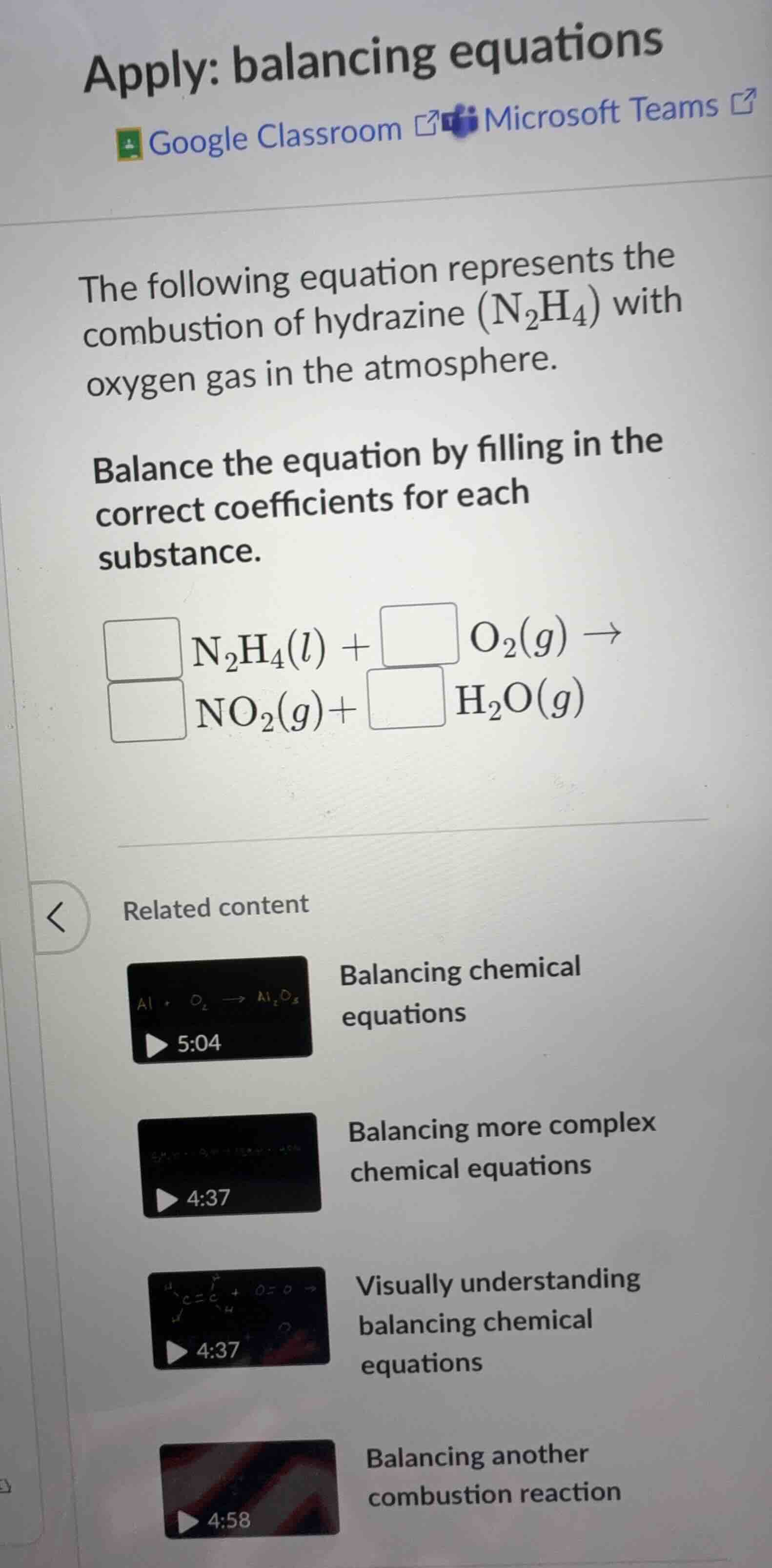
the following equation represents the combustion of hydrazine (n₂h₄) with oxygen gas in the atmosphere.
balance the equation by filling in the correct coefficients for each substance.
□ n₂h₄(l) + □ o₂(g) →
□ no₂(g)+ □ h₂o(g)
related content
balancing chemical equations
5:04
balancing more complex chemical equations
4:37
visually understanding balancing chemical equations
4:37
balancing another combustion reaction
4:58
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$1\ \text{N}_2\text{H}_4(l) + 3\ \text{O}_2(g)
ightarrow 2\ \text{NO}_2(g) + 2\ \text{H}_2\text{O}(g)$