QUESTION IMAGE
Question
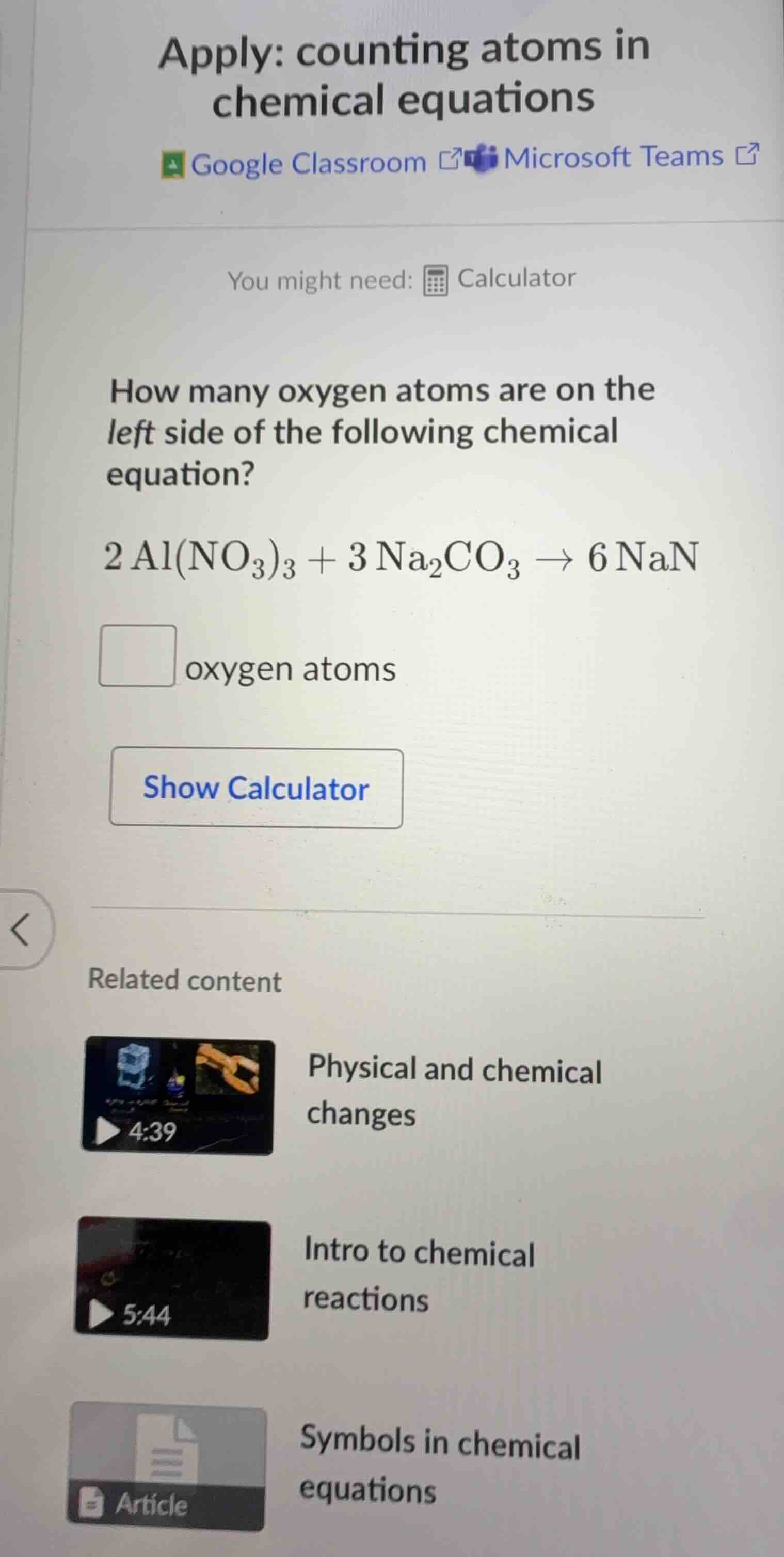
apply: counting atoms in chemical equations google classroom microsoft teams you might need: calculator how many oxygen atoms are on the left side of the following chemical equation? 2 al(no₃)₃ + 3 na₂co₃ → 6 nan oxygen atoms show calculator related content physical and chemical changes 4:39 intro to chemical reactions 5:44 symbols in chemical equations article
Step1: Analyze \( \text{Al(NO}_3\text{)}_3 \)
In \( \text{Al(NO}_3\text{)}_3 \), each formula unit has 3 \( \text{NO}_3^- \) groups, and each \( \text{NO}_3^- \) has 3 O atoms. So per \( \text{Al(NO}_3\text{)}_3 \), O atoms: \( 3\times3 = 9 \). With coefficient 2, total O from \( 2\text{Al(NO}_3\text{)}_3 \): \( 2\times9 = 18 \).
Step2: Analyze \( \text{Na}_2\text{CO}_3 \)
In \( \text{Na}_2\text{CO}_3 \), each formula unit has 3 O atoms. With coefficient 3, total O from \( 3\text{Na}_2\text{CO}_3 \): \( 3\times3 = 9 \).
Step3: Sum O atoms
Add O atoms from both compounds: \( 18 + 9 = 27 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
27