QUESTION IMAGE
Question
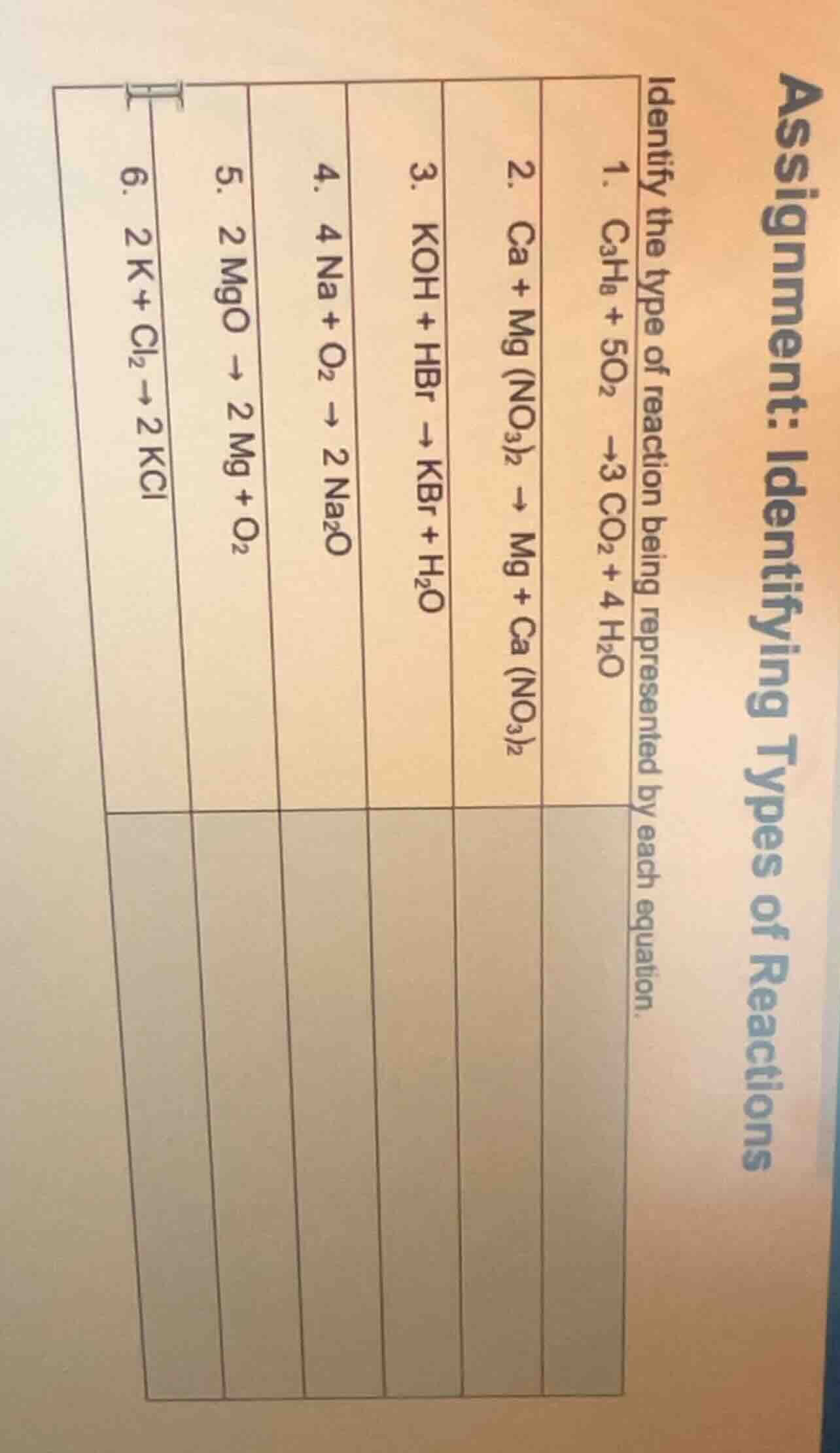
assignment: identifying types of reactions
identify the type of reaction being represented by each equation.
- $\ce{c3h8 + 5o2 -> 3co2 + 4h2o}$
- $\ce{ca + mg(no3)2 -> mg + ca(no3)2}$
- $\ce{koh + hbr -> kbr + h2o}$
- $\ce{4na + o2 -> 2na2o}$
- $\ce{2mgo -> 2mg + o2}$
- $\ce{2k + cl2 -> 2kcl}$
1. For $\boldsymbol{\ce{C3H8 + 5O2 -> 3CO2 + 4H2O}}$
Step1: Recall reaction types
Combustion reactions involve a hydrocarbon (compound with C and H) reacting with $\ce{O2}$ to produce $\ce{CO2}$ and $\ce{H2O}$. Here, $\ce{C3H8}$ (a hydrocarbon) reacts with $\ce{O2}$, forming $\ce{CO2}$ and $\ce{H2O}$.
Step2: Classify the reaction
This fits the definition of a combustion reaction.
Step1: Recall reaction types
In a single - displacement reaction, one element replaces another element in a compound. Here, $\ce{Ca}$ (a metal) replaces $\ce{Mg}$ in $\ce{Mg(NO3)2}$.
Step2: Classify the reaction
This follows the pattern of a single - displacement reaction ($\ce{A + BC -> B + AC}$, where $\ce{A = Ca}$, $\ce{B = Mg}$, $\ce{C = NO3^-}$).
Step1: Recall reaction types
A neutralization reaction (a type of double - displacement reaction) occurs when an acid ($\ce{HBr}$) reacts with a base ($\ce{KOH}$) to form a salt ($\ce{KBr}$) and water ($\ce{H2O}$). The general form of a double - displacement reaction is $\ce{AB + CD -> AD + CB}$, here $\ce{A = K}$, $\ce{B = OH}$, $\ce{C = H}$, $\ce{D = Br}$.
Step2: Classify the reaction
Since an acid and a base are reacting to form a salt and water, it is a neutralization (double - displacement) reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Combustion Reaction