QUESTION IMAGE
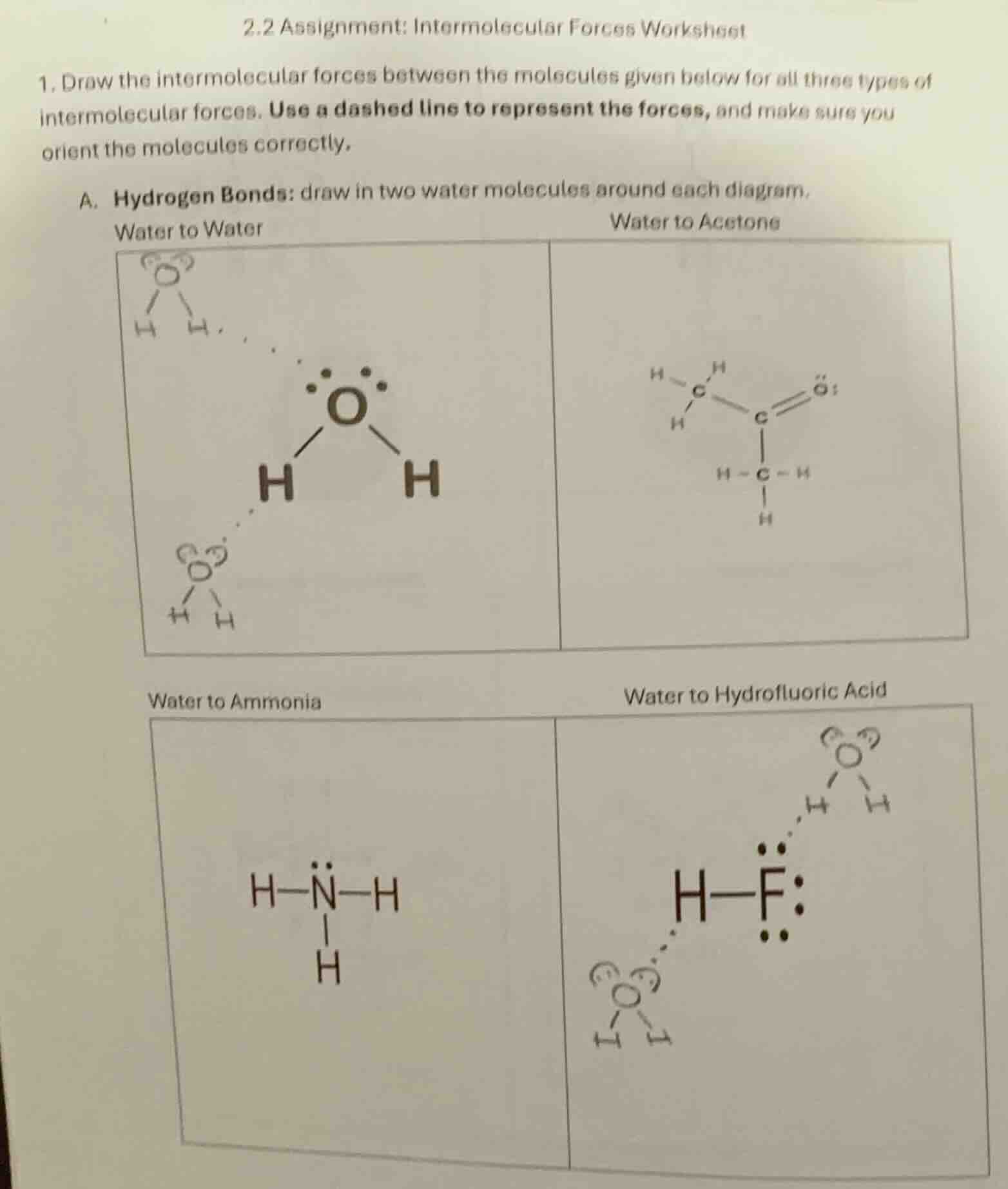
Question
2.2 assignment: intermolecular forces worksheet
- draw the intermolecular forces between the molecules given below for all three types of intermolecular forces. use a dashed line to represent the forces, and make sure you orient the molecules correctly.
a. hydrogen bonds: draw in two water molecules around each diagram.
water to water
water to acetone
water to ammonia
water to hydrofluoric acid
To draw hydrogen bonds:
- Water to Water: A water molecule (\(H_2O\)) has \(O\) (electronegative, lone pairs) and \(H\) (bonded to \(O\)). The \(H\) of one \(H_2O\) forms a dashed line to the \(O\) (with lone pairs) of another \(H_2O\). Orient the second \(H_2O\) so its \(O\) faces the \(H\) of the first, and add a second \(H_2O\) similarly (e.g., one \(H\) of the new \(H_2O\) bonds to the \(O\) of the original, and vice versa).
- Water to Acetone (\(CH_3COCH_3\)): Acetone has a carbonyl (\(C=O\)) group. The \(O\) in \(C=O\) has lone pairs. The \(H\) of \(H_2O\) forms a dashed line to the \(O\) of acetone’s \(C=O\). Draw \(H_2O\) so its \(H\) points to acetone’s \(O\), and add a second \(H_2O\) (e.g., another \(H\) of \(H_2O\) bonds to acetone’s \(O\) or the first \(H_2O\)’s \(O\)).
- Water to Ammonia (\(NH_3\)): Ammonia has \(N\) with lone pairs. The \(H\) of \(H_2O\) forms a dashed line to the \(N\) of \(NH_3\). Orient \(H_2O\) so its \(H\) faces \(NH_3\)’s \(N\), and add a second \(H_2O\) (e.g., \(H\) of new \(H_2O\) bonds to \(NH_3\)’s \(N\) or the first \(H_2O\)’s \(O\)).
- Water to Hydrofluoric Acid (\(HF\)): \(HF\) has \(F\) (electronegative, lone pairs) and \(H\) (bonded to \(F\)). The \(H\) of \(H_2O\) bonds to \(F\) of \(HF\), and the \(H\) of \(HF\) bonds to \(O\) of \(H_2O\). Draw \(H_2O\) so its \(O\) faces \(HF\)’s \(H\), and add a second \(H_2O\) (e.g., \(H\) of new \(H_2O\) bonds to \(HF\)’s \(F\) or the first \(H_2O\)’s \(O\)).
Key: Hydrogen bonds form between a hydrogen bonded to \(F\), \(O\), or \(N\) (in one molecule) and a lone pair on \(F\), \(O\), or \(N\) (in another molecule). Use dashed lines for these interactions and ensure molecular orientation (electronegative atom with lone pairs faces the hydrogen).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Diagrams should be drawn as follows, with dashed lines for hydrogen bonds and correct molecular orientation:
- Water to Water: Two \(H_2O\) molecules with \(H\)-to-\(O\) dashed lines (e.g., \(H\) of first \(H_2O\) to \(O\) of second, and \(H\) of second to \(O\) of first).
- Water to Acetone: \(H_2O\)’s \(H\) to acetone’s \(C=O\) \(O\) (dashed line), plus a second \(H_2O\) with similar bonding.
- Water to Ammonia: \(H_2O\)’s \(H\) to \(NH_3\)’s \(N\) (dashed line), plus a second \(H_2O\) with similar bonding.
- Water to Hydrofluoric Acid: \(H_2O\)’s \(H\) to \(HF\)’s \(F\), and \(HF\)’s \(H\) to \(H_2O\)’s \(O\) (dashed lines), plus a second \(H_2O\) with similar bonding.
)
(Note: Since this is a drawing task, the answer describes the bonding pattern. For actual diagrams, sketch molecules with dashed lines between the relevant atoms, ensuring correct orientation for hydrogen bonding.)