QUESTION IMAGE
Question
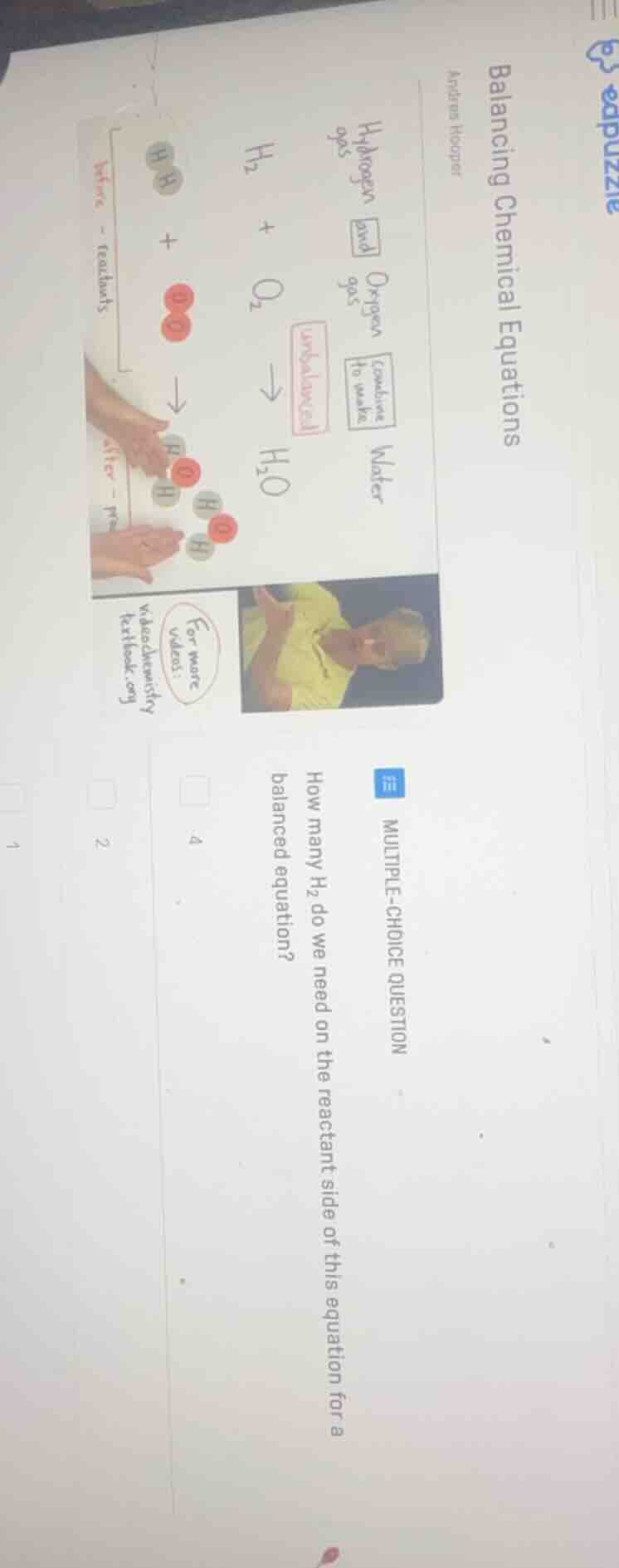
balancing chemical equations
multiple-choice question
how many h₂ do we need on the reactant side of this equation for a balanced equation?
4
2
1
Step1: Identify unbalanced equation
$H_2 + O_2
ightarrow H_2O$
Step2: Balance oxygen atoms
Add coefficient 2 to $H_2O$:
$H_2 + O_2
ightarrow 2H_2O$
Step3: Balance hydrogen atoms
Add coefficient 2 to $H_2$:
$2H_2 + O_2
ightarrow 2H_2O$
Step4: Verify atom counts
Reactants: 4 H, 2 O; Products: 4 H, 2 O. Equation is balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2