QUESTION IMAGE
Question
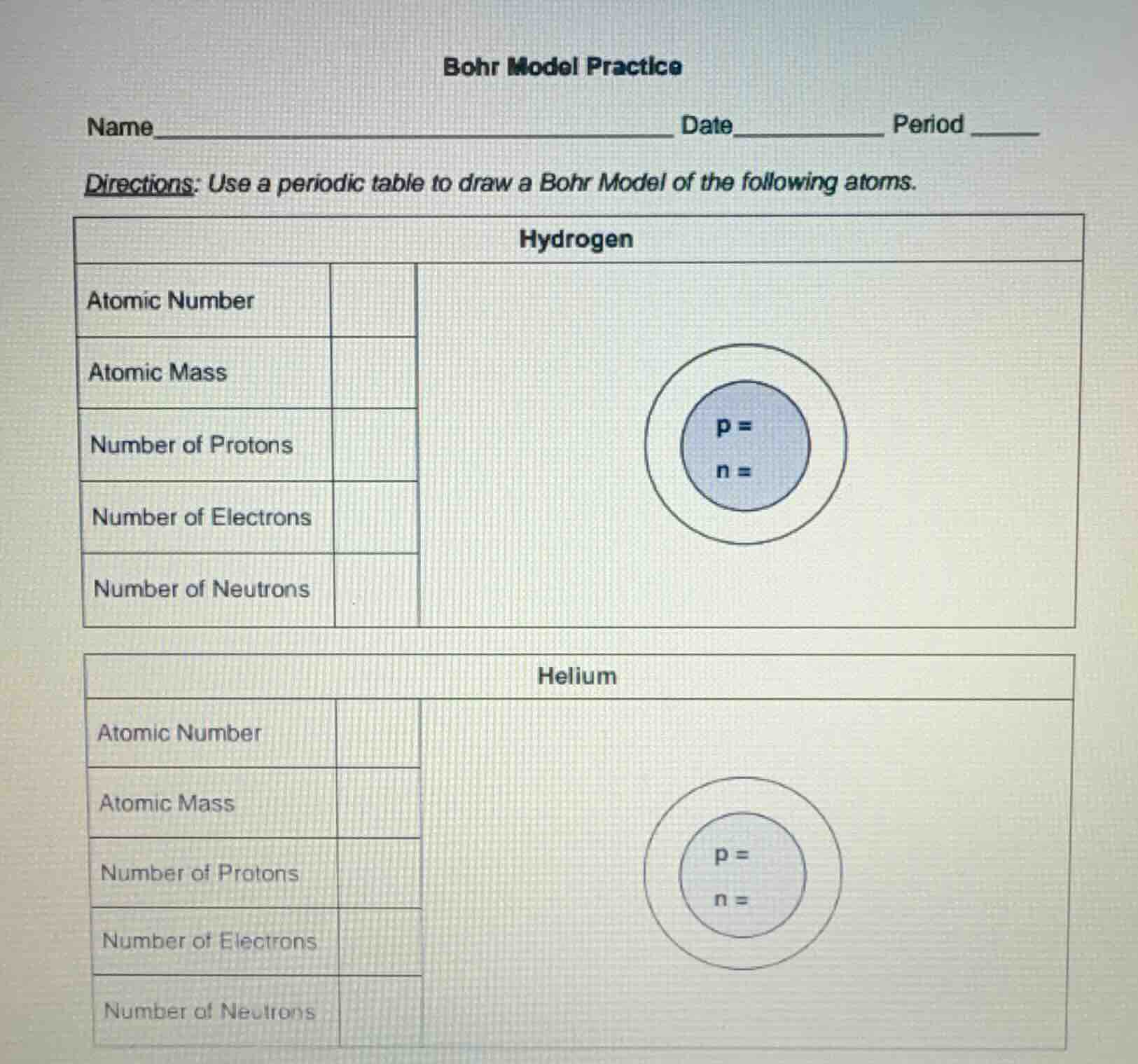
bohr model practice
name______________________ date______ period ____
directions: use a periodic table to draw a bohr model of the following atoms.
hydrogen
atomic number
atomic mass
number of protons
number of electrons
number of neutrons
p =
n =
helium
atomic number
atomic mass
number of protons
number of electrons
number of neutrons
p =
n =
Step1: Find Hydrogen's atomic data
From periodic table:
Atomic Number = 1
Atomic Mass = 1.008 ≈ 1
Protons = Atomic Number = 1
Electrons = Protons = 1
Neutrons = Atomic Mass - Protons = $1 - 1 = 0$
Step2: Find Helium's atomic data
From periodic table:
Atomic Number = 2
Atomic Mass = 4.003 ≈ 4
Protons = Atomic Number = 2
Electrons = Protons = 2
Neutrons = Atomic Mass - Protons = $4 - 2 = 2$
Step3: Summarize Bohr model details
Hydrogen: 1 proton, 0 neutrons in nucleus; 1 electron in 1st energy level.
Helium: 2 protons, 2 neutrons in nucleus; 2 electrons in 1st energy level.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Hydrogen
| Property | Value | Bohr Model Details |
|---|---|---|
| Atomic Mass | 1.008 | |
| Number of Protons | 1 | |
| Number of Electrons | 1 | |
| Number of Neutrons | 0 |
Helium
| Property | Value | Bohr Model Details |
|---|---|---|
| Atomic Mass | 4.003 | |
| Number of Protons | 2 | |
| Number of Electrons | 2 | |
| Number of Neutrons | 2 |