QUESTION IMAGE
Question
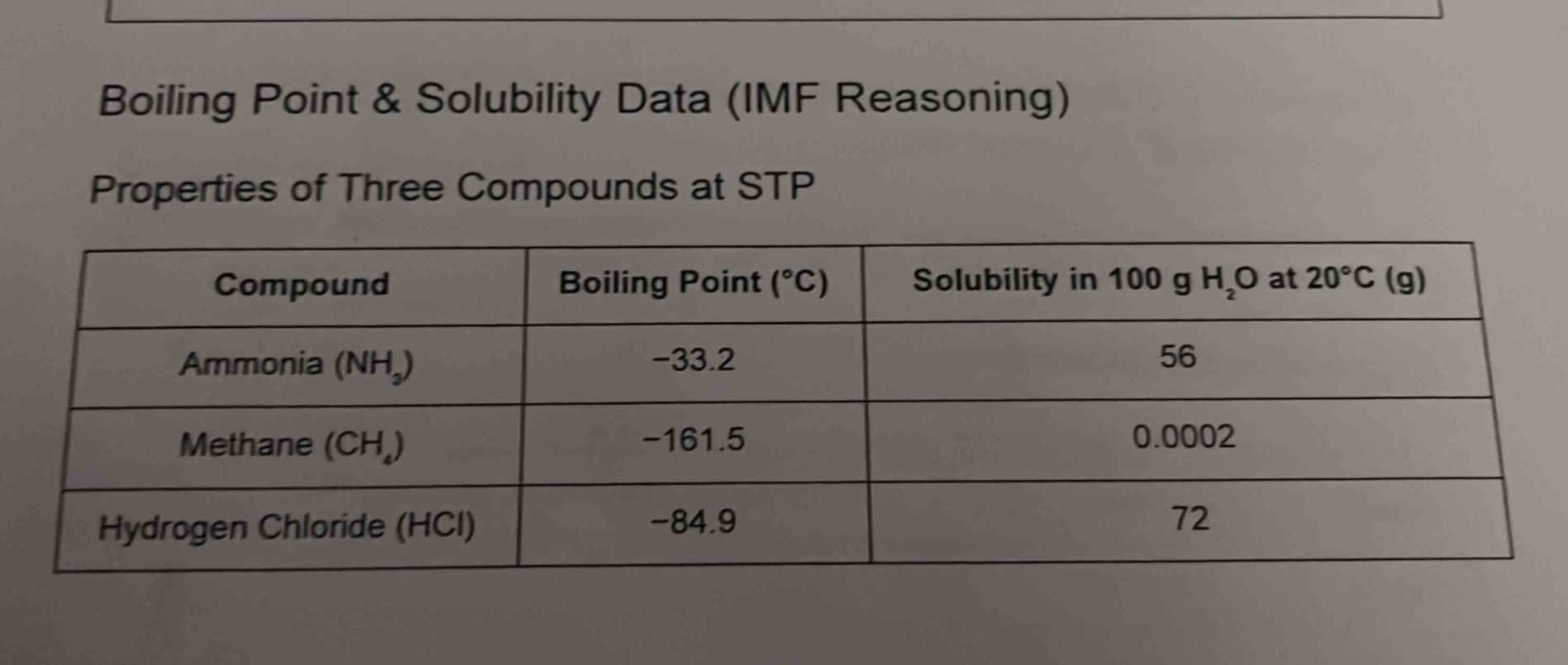
boiling point & solubility data (imf reasoning)
properties of three compounds at stp
| compound | boiling point (°c) | solubility in 100 g h₂o at 20°c (g) |
|---|---|---|
| methane (ch₄) | -161.5 | 0.0002 |
| hydrogen chloride (hcl) | -84.9 | 72 |
Since the problem (presumably about analyzing boiling points and solubilities using intermolecular forces) isn't fully stated, but if we assume a common question like "Which compound has the highest boiling point?" or "Explain solubility differences using IMF":
For a question like "Which compound has the highest boiling point?"
Step1: Compare boiling point values
Ammonia: $-33.2^\circ\text{C}$, Methane: $-161.5^\circ\text{C}$, Hydrogen Chloride: $-84.9^\circ\text{C}$.
Step2: Identify the highest value
Among $-33.2$, $-161.5$, and $-84.9$, $-33.2$ is the highest (closest to 0). So ammonia has the highest boiling point.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Ammonia ($\ce{NH_3}$) has the highest boiling point with a boiling point of $-33.2^\circ\text{C}$.
If the question was about solubility, the highest solubility is 72 g (Hydrogen Chloride), followed by 56 g (Ammonia), then 0.0002 g (Methane). The reasoning would involve IMF: $\ce{NH_3}$ and $\ce{HCl}$ can form hydrogen bonds or dipole - dipole interactions with water (polar), while $\ce{CH_4}$ is non - polar, so it has low solubility. $\ce{HCl}$ has higher solubility than $\ce{NH_3}$ maybe due to stronger dipole - dipole or ion - dipole (when dissociated) interactions.