QUESTION IMAGE
Question
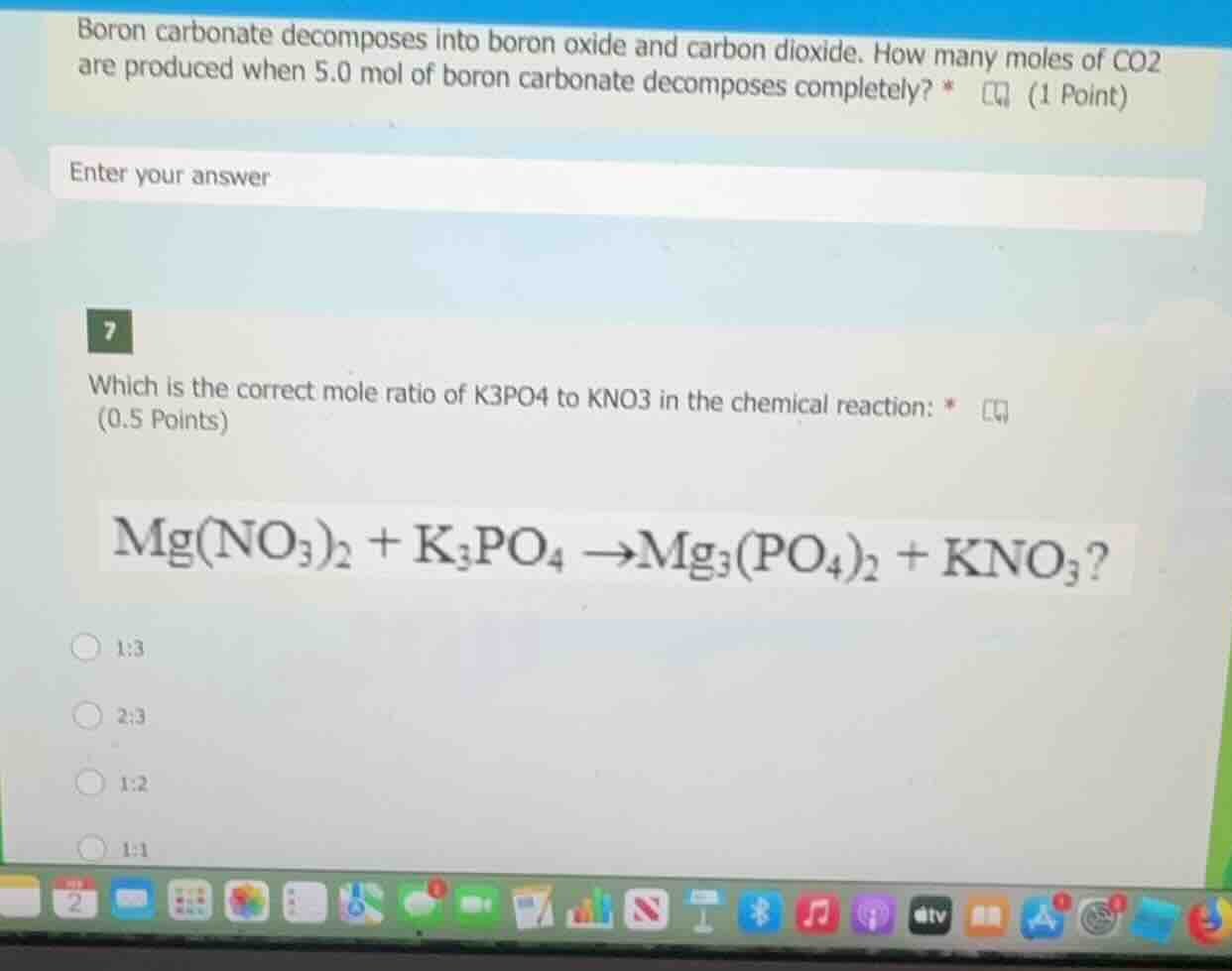
boron carbonate decomposes into boron oxide and carbon dioxide. how many moles of co2 are produced when 5.0 mol of boron carbonate decomposes completely? (1 point) enter your answer 7 which is the correct mole ratio of k3po4 to kno3 in the chemical reaction: (0.5 points) mg(no3)2 + k3po4 →mg3(po4)2 + kno3? 1:3 2:3 1:2 1:1
First Question (Boron Carbonate Decomposition)
Step1: Write the balanced equation
The formula for boron carbonate is \( B_2(CO_3)_3 \), boron oxide is \( B_2O_3 \), and carbon dioxide is \( CO_2 \). The decomposition reaction is:
\( B_2(CO_3)_3
ightarrow B_2O_3 + 3CO_2 \) (balanced by ensuring C and O are balanced: 3 \( CO_2 \) from 3 \( CO_3^{2-} \) groups).
Step2: Determine mole ratio
From the balanced equation, 1 mol of \( B_2(CO_3)_3 \) produces 3 mol of \( CO_2 \).
Step3: Calculate moles of \( CO_2 \)
Given 5.0 mol of \( B_2(CO_3)_3 \), use the ratio:
Moles of \( CO_2 = 5.0 \, \text{mol} \times \frac{3 \, \text{mol} \, CO_2}{1 \, \text{mol} \, B_2(CO_3)_3} = 15 \, \text{mol} \).
Step1: Balance the chemical equation
The unbalanced equation is:
\( Mg(NO_3)_2 + K_3PO_4
ightarrow Mg_3(PO_4)_2 + KNO_3 \)
- Balance Mg: 3 \( Mg(NO_3)_2 \) (since product has 3 Mg).
- Balance \( PO_4^{3-} \): 2 \( K_3PO_4 \) (since product has 2 \( PO_4^{3-} \)).
- Balance \( NO_3^- \): 6 \( KNO_3 \) (from 3 \( Mg(NO_3)_2 \), which has 6 \( NO_3^- \)).
- Balance K: 2 \( K_3PO_4 \) gives 6 K, which matches 6 \( KNO_3 \).
Balanced equation:
\( 3Mg(NO_3)_2 + 2K_3PO_4
ightarrow Mg_3(PO_4)_2 + 6KNO_3 \)
Step2: Find mole ratio of \( K_3PO_4 \) to \( KNO_3 \)
From the balanced equation, 2 mol of \( K_3PO_4 \) produces 6 mol of \( KNO_3 \). Simplify the ratio:
\( \frac{\text{Moles of } K_3PO_4}{\text{Moles of } KNO_3} = \frac{2}{6} = \frac{1}{3} \), so the ratio is \( 1:3 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
15