QUESTION IMAGE
Question
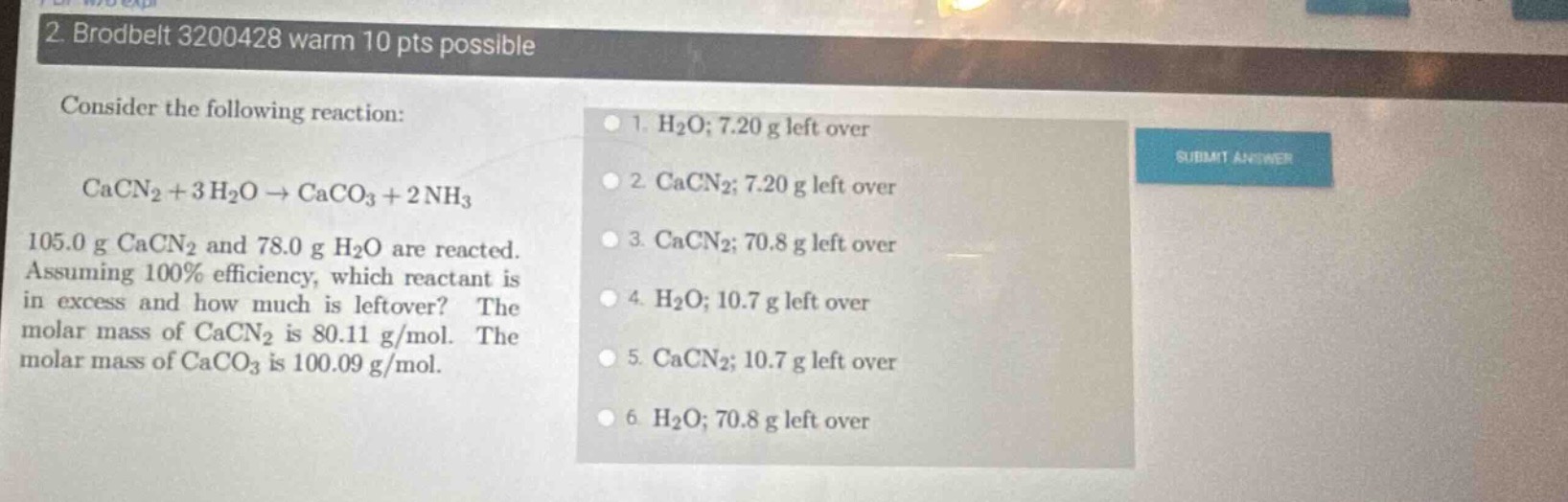
- brodbelt 3200428 warm 10 pts possible
consider the following reaction:
\\(\ce{cacn2 + 3 h2o -> caco3 + 2 nh3}\\)
105.0 g \\(\ce{cacn2}\\) and 78.0 g \\(\ce{h2o}\\) are reacted. assuming 100% efficiency, which reactant is in excess and how much is leftover? the molar mass of \\(\ce{cacn2}\\) is 80.11 g/mol. the molar mass of \\(\ce{caco3}\\) is 100.09 g/mol.
- \\(\ce{h2o}\\); 7.20 g left over
- \\(\ce{cacn2}\\); 7.20 g left over
- \\(\ce{cacn2}\\); 70.8 g left over
- \\(\ce{h2o}\\); 10.7 g left over
- \\(\ce{cacn2}\\); 10.7 g left over
6 \\(\ce{h2o}\\); 70.8 g left over
Step1: Calculate moles of \( \text{CaCN}_2 \)
Moles of \( \text{CaCN}_2 = \frac{\text{mass}}{\text{molar mass}} = \frac{105.0\ \text{g}}{80.11\ \text{g/mol}} \approx 1.311\ \text{mol} \)
Step2: Calculate moles of \( \text{H}_2\text{O} \)
Molar mass of \( \text{H}_2\text{O} = 18.02\ \text{g/mol} \)
Moles of \( \text{H}_2\text{O} = \frac{78.0\ \text{g}}{18.02\ \text{g/mol}} \approx 4.328\ \text{mol} \)
Step3: Determine stoichiometric ratio
From reaction: \( 1\ \text{mol}\ \text{CaCN}_2 : 3\ \text{mol}\ \text{H}_2\text{O} \)
Moles of \( \text{H}_2\text{O} \) required for \( 1.311\ \text{mol}\ \text{CaCN}_2 = 1.311 \times 3 = 3.933\ \text{mol} \)
Step4: Identify limiting and excess reactant
Available \( \text{H}_2\text{O} = 4.328\ \text{mol} \), required \( = 3.933\ \text{mol} \). So \( \text{H}_2\text{O} \) is excess? Wait, no: Wait, check again. Wait, moles of \( \text{CaCN}_2 = 1.311 \), moles of \( \text{H}_2\text{O} \) needed for \( \text{CaCN}_2 \): \( 1.311 \times 3 = 3.933 \). Available \( \text{H}_2\text{O} = 4.328 \), so \( \text{H}_2\text{O} \) is in excess? Wait, no, wait: Wait, maybe I mixed up. Wait, let's check moles of \( \text{H}_2\text{O} \) and see how much \( \text{CaCN}_2 \) it can react with. Moles of \( \text{H}_2\text{O} = 4.328 \), so moles of \( \text{CaCN}_2 \) that can react with \( \text{H}_2\text{O} = \frac{4.328}{3} \approx 1.443\ \text{mol} \). But we have only \( 1.311\ \text{mol}\ \text{CaCN}_2 \), so \( \text{CaCN}_2 \) is limiting? Wait, no, that can't be. Wait, no: The reaction is \( \text{CaCN}_2 + 3\ \text{H}_2\text{O}
ightarrow \dots \). So 1 mol \( \text{CaCN}_2 \) reacts with 3 mol \( \text{H}_2\text{O} \). So if we have 1.311 mol \( \text{CaCN}_2 \), we need 3.933 mol \( \text{H}_2\text{O} \). We have 4.328 mol \( \text{H}_2\text{O} \), so \( \text{H}_2\text{O} \) is in excess? Wait, but the options have \( \text{CaCN}_2 \) or \( \text{H}_2\text{O} \) as excess. Wait, maybe I made a mistake. Wait, let's recalculate.
Wait, mass of \( \text{CaCN}_2 = 105.0\ \text{g} \), molar mass 80.11 g/mol: moles = 105 / 80.11 ≈ 1.3107 mol.
Mass of \( \text{H}_2\text{O} = 78.0\ \text{g} \), molar mass 18.015 g/mol (more accurate: 18.015). So moles = 78.0 / 18.015 ≈ 4.330 mol.
Stoichiometric ratio: 1 mol \( \text{CaCN}_2 \) needs 3 mol \( \text{H}_2\text{O} \). So moles of \( \text{H}_2\text{O} \) needed for \( \text{CaCN}_2 \): 1.3107 * 3 = 3.9321 mol.
Available \( \text{H}_2\text{O} \): 4.330 mol. So excess \( \text{H}_2\text{O} \) moles = 4.330 - 3.9321 = 0.3979 mol.
Mass of excess \( \text{H}_2\text{O} \): 0.3979 mol * 18.015 g/mol ≈ 7.16 g ≈ 7.20 g. Wait, but that's option 1: \( \text{H}_2\text{O} \); 7.20 g left over. Wait, but wait, maybe I mixed up limiting reactant. Wait, no: If \( \text{CaCN}_2 \) is limiting, then \( \text{H}_2\text{O} \) is excess. So the excess is \( \text{H}_2\text{O} \), and mass left over is ~7.20 g. So option 1.
Wait, let's check again. Moles of \( \text{CaCN}_2 = 105 / 80.11 ≈ 1.3107 \). Moles of \( \text{H}_2\text{O} \) needed: 1.3107 3 = 3.9321. Moles of \( \text{H}_2\text{O} \) available: 78 / 18.015 ≈ 4.330. Excess moles: 4.330 - 3.9321 = 0.3979. Mass: 0.3979 18.015 ≈ 7.16 ≈ 7.20 g. So option 1: \( \text{H}_2\text{O} \); 7.20 g left over.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- \( \text{H}_2\text{O} \); 7.20 g left over