QUESTION IMAGE
Question
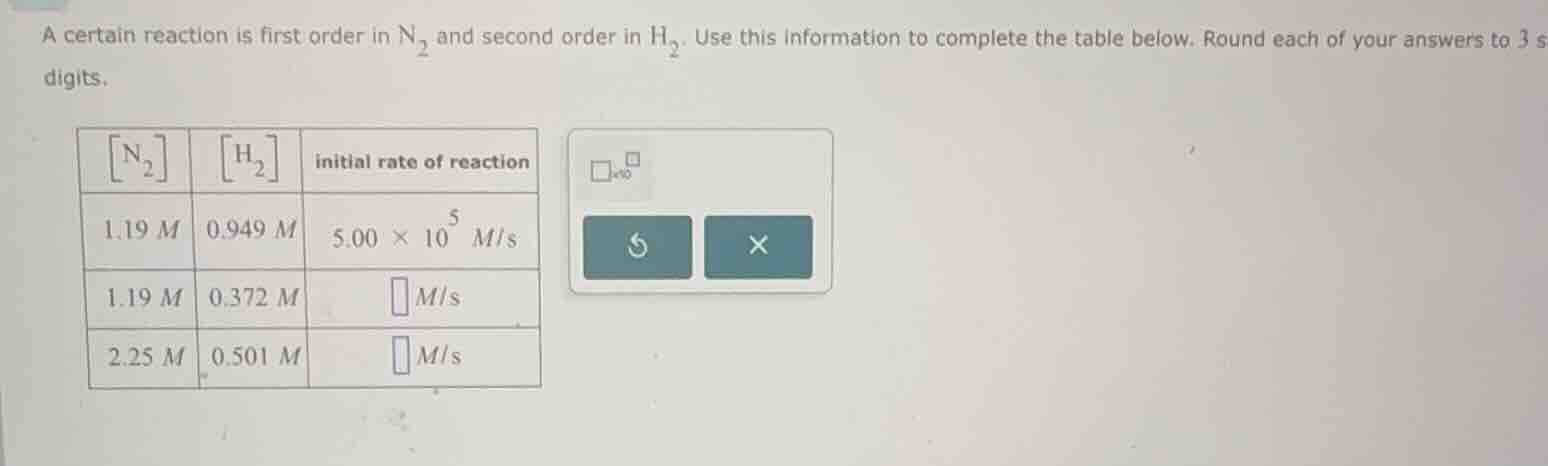
a certain reaction is first order in $ce{n_{2}}$ and second order in $ce{h_{2}}$. use this information to complete the table below. round each of your answers to 3 significant digits.
| $ce{n_{2}}$ | $ce{h_{2}}$ | initial rate of reaction | ---- | ---- | ---- | 1.19 $m$ | 0.949 $m$ | $5.00 \times 10^{5}$ $m/s$ | 1.19 $m$ | 0.372 $m$ | $square$ $m/s$ | 2.25 $m$ | 0.501 $m$ | $square$ $m/s$ |
Step1: Write rate law equation
The reaction is first-order in $\text{N}_2$ and second-order in $\text{H}_2$, so the rate law is:
$$\text{Rate} = k[\text{N}_2][\text{H}_2]^2$$
Step2: Solve for rate constant $k$
Use the first row of data to find $k$:
$$k = \frac{\text{Rate}}{[\text{N}_2][\text{H}_2]^2} = \frac{5.00 \times 10^5}{(1.19)(0.949)^2}$$
Calculate denominator: $(1.19)(0.949)^2 = 1.19 \times 0.900601 = 1.07171519$
$$k = \frac{5.00 \times 10^5}{1.07171519} \approx 4.665 \times 10^5 \ M^{-2}s^{-1}$$
Step3: Calculate rate for row 2
$[\text{N}_2]=1.19\ M$, $[\text{H}_2]=0.372\ M$:
$$\text{Rate} = 4.665 \times 10^5 \times 1.19 \times (0.372)^2$$
Calculate $(0.372)^2 = 0.138384$, then $1.19 \times 0.138384 = 0.16467696$
$$\text{Rate} = 4.665 \times 10^5 \times 0.16467696 \approx 7.68 \times 10^4$$
Step4: Calculate rate for row 3
$[\text{N}_2]=2.25\ M$, $[\text{H}_2]=0.501\ M$:
$$\text{Rate} = 4.665 \times 10^5 \times 2.25 \times (0.501)^2$$
Calculate $(0.501)^2 = 0.251001$, then $2.25 \times 0.251001 = 0.56475225$
$$\text{Rate} = 4.665 \times 10^5 \times 0.56475225 \approx 2.63 \times 10^5$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Second row initial rate: $\boldsymbol{7.68 \times 10^4\ M/s}$
Third row initial rate: $\boldsymbol{2.63 \times 10^5\ M/s}$