QUESTION IMAGE
Question
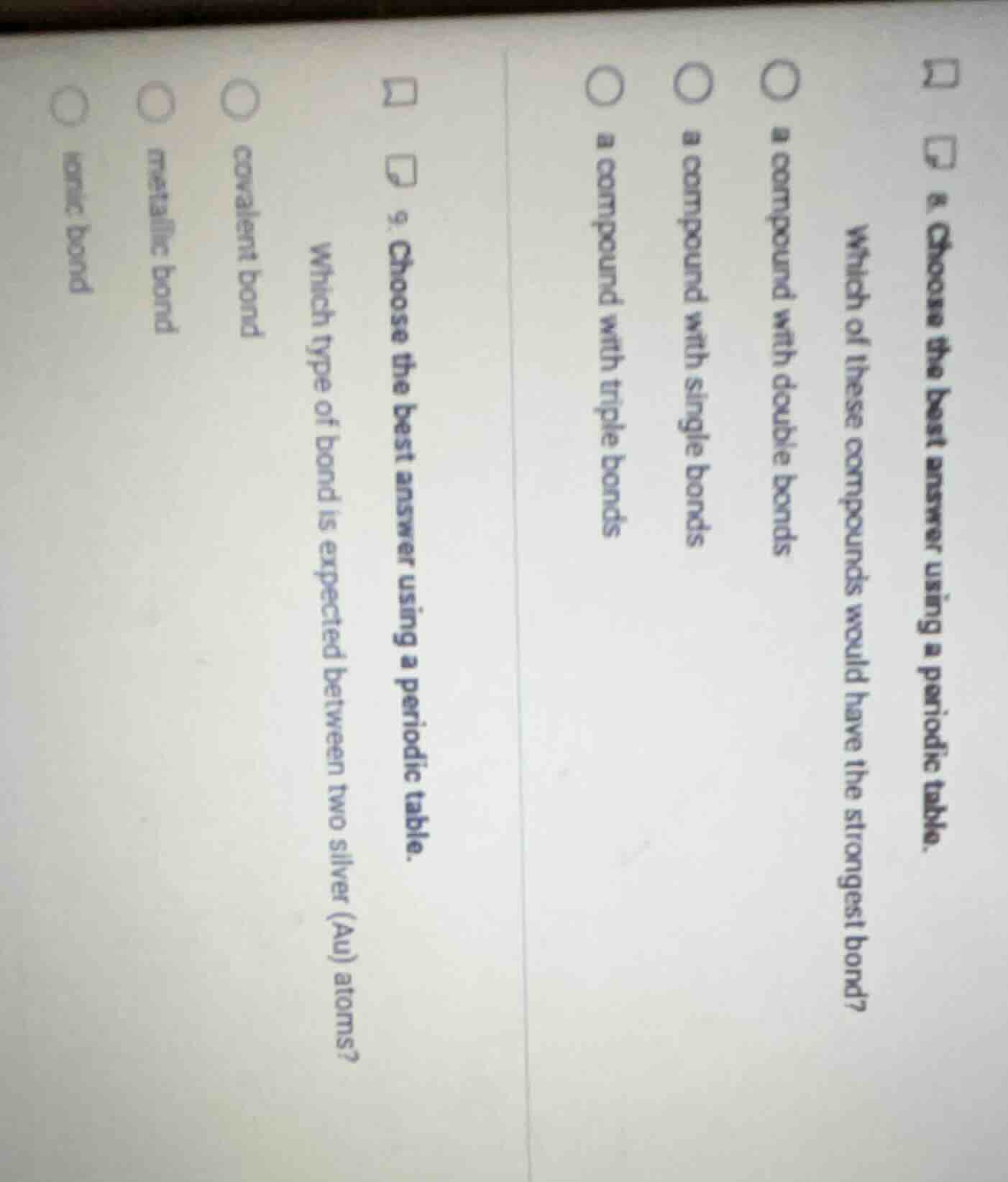
- choose the best answer using a periodic table. which of these compounds would have the strongest bond? a compound with double bonds a compound with single bonds a compound with triple bonds 9. choose the best answer using a periodic table. which type of bond is expected between two silver (au) atoms? covalent bond metallic bond ionic bond
Step1: Analyze bond strength trend
Bond strength increases with the number of shared electron pairs: triple bonds have more shared pairs than double, which have more than single.
Step2: Identify strongest bond type
Triple bonds have the highest bond dissociation energy among the options.
Step3: Classify bond between Au atoms
Silver (Au, actually gold) is a metal; metals form metallic bonds between atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- a compound with triple bonds
- metallic bond