QUESTION IMAGE
Question
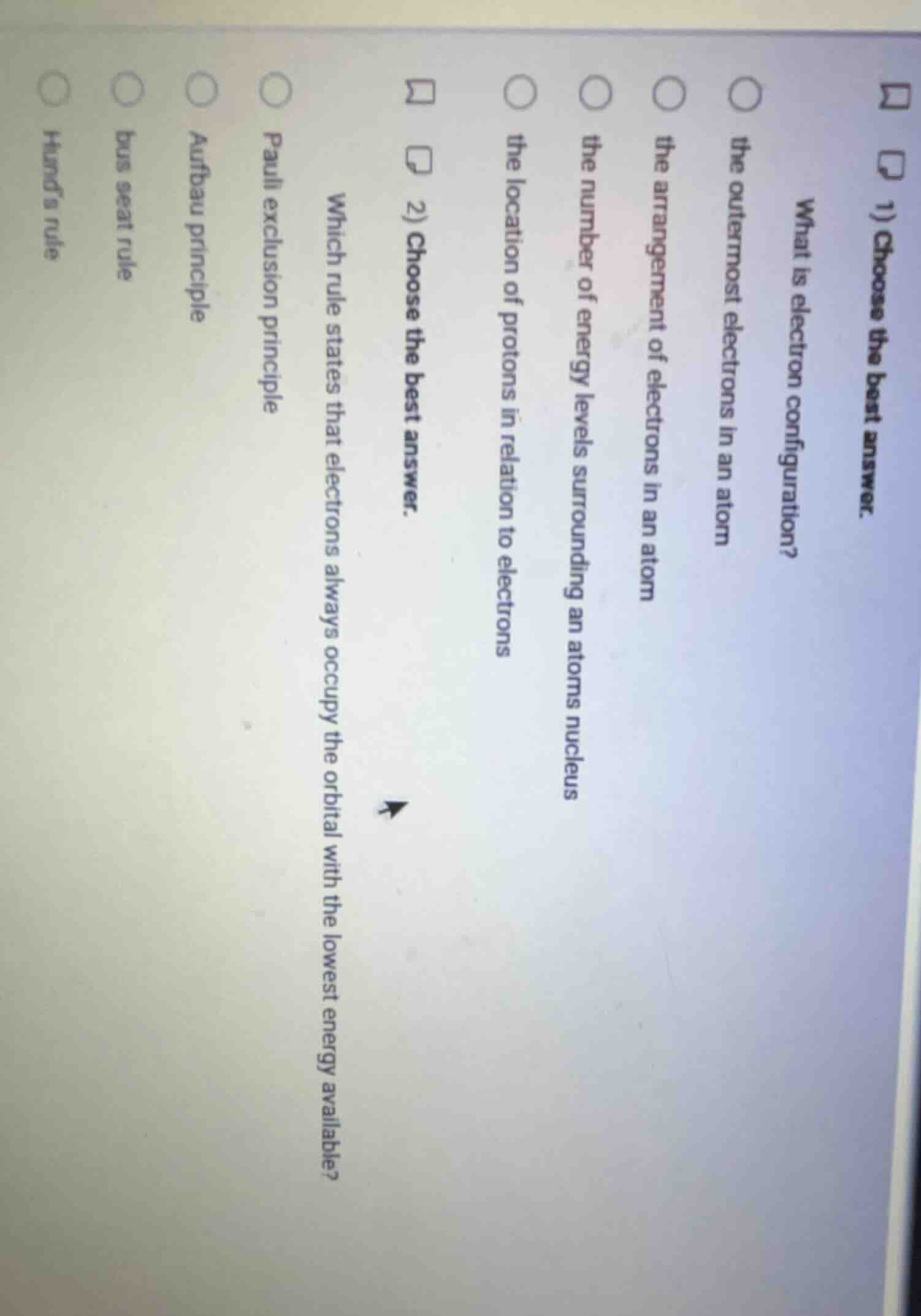
- choose the best answer. what is electron configuration? the outermost electrons in an atom the arrangement of electrons in an atom the number of energy levels surrounding an atoms nucleus the location of protons in relation to electrons 2) choose the best answer. which rule states that electrons always occupy the orbital with the lowest energy available? pauli exclusion principle aufbau principle bus seat rule hunds rule
Brief Explanations
- Electron configuration is defined as the specific arrangement of all electrons in an atom across energy levels, subshells, and orbitals.
- The rule stating electrons fill the lowest energy orbital first is the Aufbau principle; the other options describe different electron behavior rules or a non-scientific distractor.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- the arrangement of electrons in an atom
- Aufbau principle