QUESTION IMAGE
Question
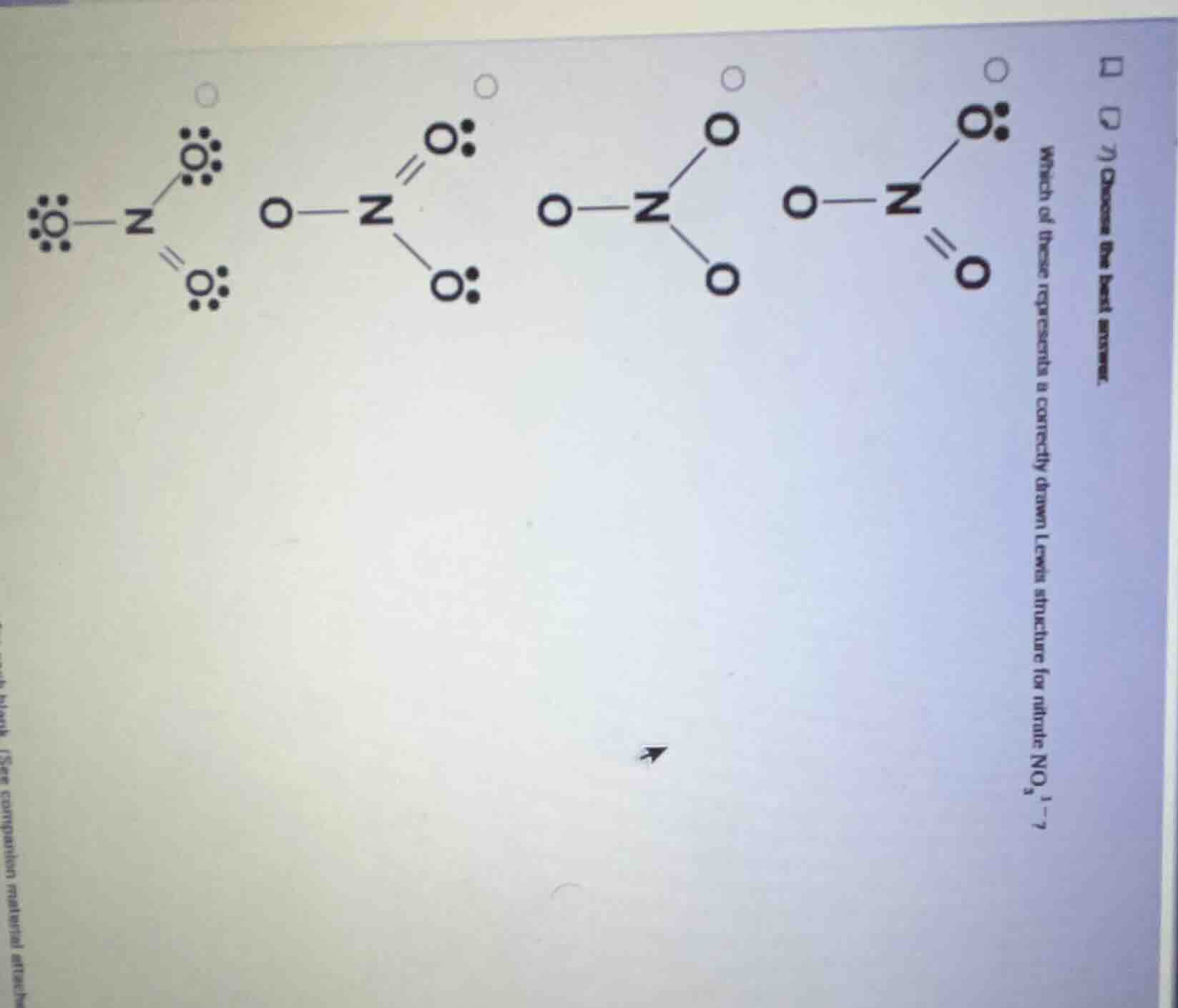
- choose the best answer. which of these represents a correctly drawn lewis structure for nitrate no₃⁻?
Brief Explanations
- Calculate total valence electrons for $\text{NO}_3^-$: Nitrogen has 5, each oxygen has 6, plus 1 for the negative charge: $5 + (3 \times 6) + 1 = 24$ total electrons.
- Nitrogen is the central atom. A valid Lewis structure for nitrate has one N=O double bond and two N-O single bonds (resonance structures), with each oxygen having a full octet (8 electrons: bonding pairs + lone pairs).
- Analyze options:
- Option 1: Incorrect, nitrogen has more than 8 electrons.
- Option 2: Incorrect, does not account for all 24 electrons and octets.
- Option 3: Incorrect, only shows 6 bonding electrons (3 single bonds) with no lone pairs, missing 18 electrons.
- Option 4: Correct, has 1 double bond, 2 single bonds, each oxygen has 3 lone pairs (6 electrons) plus bonding electrons to complete an octet, and all 24 valence electrons are accounted for.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The fourth option (rightmost): $\text{O}-\text{N}(=\text{O})-\ddot{\text{O}}:$ (with three lone pairs on the single-bonded oxygens and two lone pairs on the double-bonded oxygen)