QUESTION IMAGE
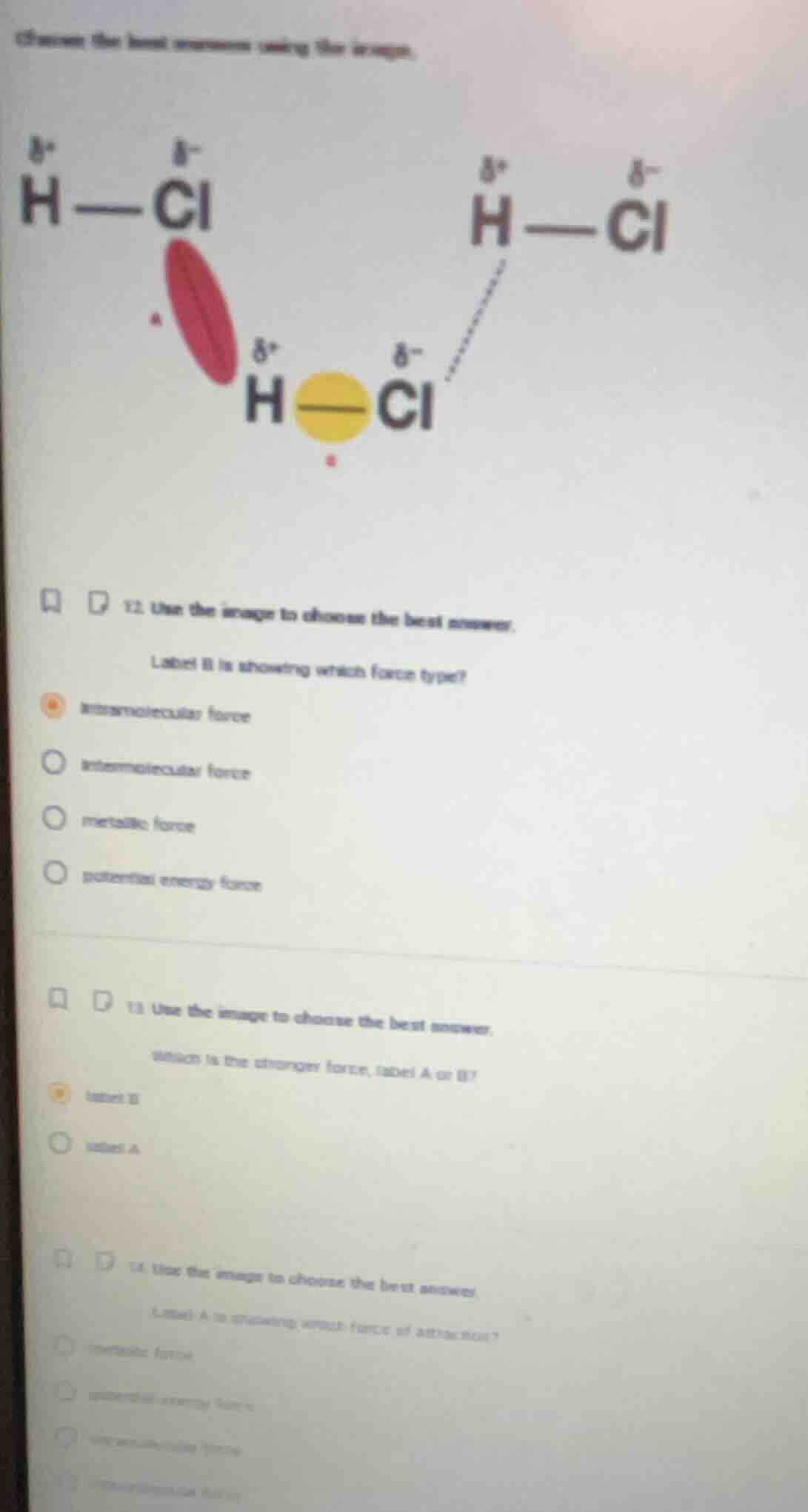
Question
choose the best answers using the image.
- use the image to choose the best answer.
label b is showing which force type?
intramolecular force
intermolecular force
metallic force
potential energy force
- use the image to choose the best answer.
which is the stronger force, label a or b?
label b
label a
- use the image to choose the best answer.
label a is showing which force of attraction?
metallic force
potential energy force
intermolecular force
intramolecular force
Brief Explanations
- For Q2: Label B is the solid bond within a single HCl molecule, which holds the atoms of the molecule together, so it is an intramolecular force.
- For Q3: Intramolecular forces (Label B) are stronger than intermolecular forces (Label A), as they involve direct bonding between atoms rather than weaker attractions between molecules.
- For Q4: Label A is the dashed attraction between the partially positive H of one HCl molecule and the partially negative Cl of another, which is an intermolecular force (specifically dipole-dipole interaction, a type of intermolecular force).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Q2: A. intramolecular force
Q3: A. Label B
Q4: C. intermolecular force