QUESTION IMAGE
Question
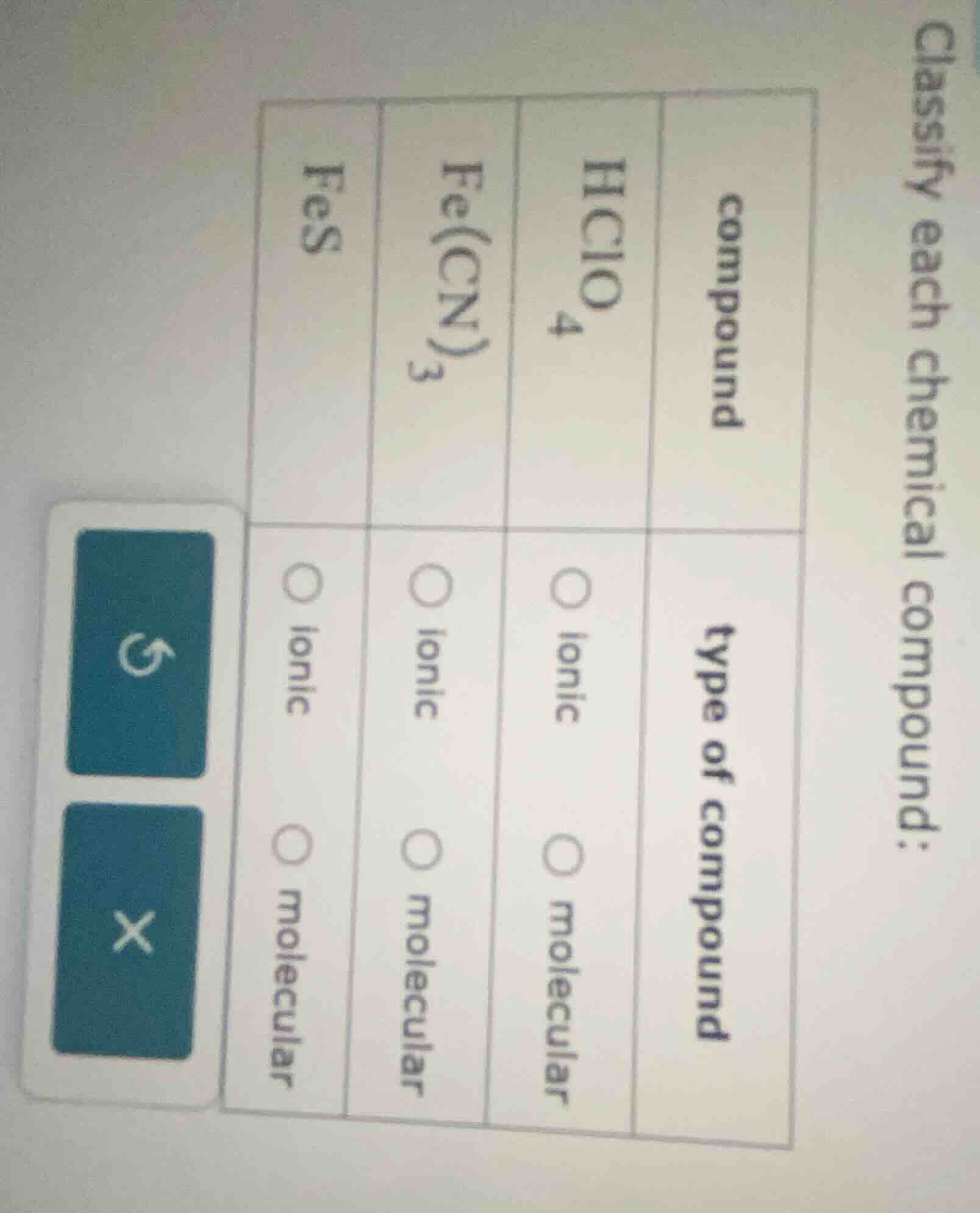
classify each chemical compound:
| compound | type of compound |
|---|---|
| fe(cn)₃ | 〇 ionic 〇 molecular |
| fes | 〇 ionic 〇 molecular |
Step1: Analyze \( \text{HClO}_4 \)
\( \text{HClO}_4 \) (perchloric acid) consists of \( \text{H}^+ \) and \( \text{ClO}_4^- \) ions. Ionic compounds are formed from ions (metal - nonmetal or polyatomic ions with ions). So \( \text{HClO}_4 \) is ionic.
Step2: Analyze \( \text{Fe(CN)}_3 \)
\( \text{Fe(CN)}_3 \) has \( \text{Fe}^{3+} \) and \( \text{CN}^- \) (cyanide, a polyatomic ion) ions. The combination of a metal ion (\( \text{Fe}^{3+} \)) and polyatomic ions (\( \text{CN}^- \)) forms an ionic compound. So \( \text{Fe(CN)}_3 \) is ionic.
Step3: Analyze \( \text{FeS} \)
\( \text{FeS} \) is formed from \( \text{Fe}^{2+} \) (iron(II) ion) and \( \text{S}^{2 - } \) (sulfide ion). Metal (Fe) and non - metal (S) form an ionic compound through the transfer of electrons. So \( \text{FeS} \) is ionic.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- \( \text{HClO}_4 \): ionic
- \( \text{Fe(CN)}_3 \): ionic
- \( \text{FeS} \): ionic