QUESTION IMAGE
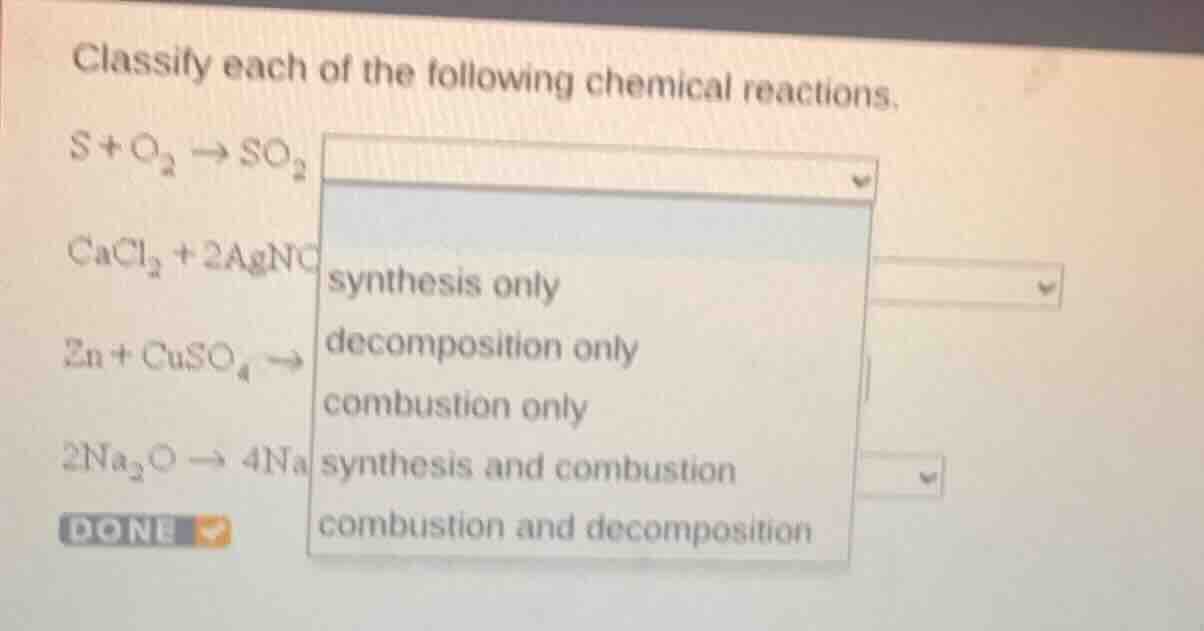
Question
classify each of the following chemical reactions.
$\ce{s + o_{2} \
ightarrow so_{2}}$
$\ce{cacl_{2} + 2agno}$
$\ce{zn + cuso_{4} \
ightarrow}$
$\ce{2na_{2}o \
ightarrow 4na}$
done
synthesis only
decomposition only
combustion only
synthesis and combustion
combustion and decomposition
For the reaction $\boldsymbol{\ce{S + O_{2}
ightarrow SO_{2}}}$:
Step 1: Recall reaction types
Synthesis reaction: A + B → AB (combination of substances). Combustion: Reaction with $\ce{O_{2}}$, often forming oxides, and can be synthesis if it's A + $\ce{O_{2}}$ → AO (type of synthesis).
Here, $\ce{S}$ and $\ce{O_{2}}$ combine to form $\ce{SO_{2}}$ (synthesis: S + O₂ → SO₂), and since it involves $\ce{O_{2}}$ (combustion of S), it's both synthesis and combustion.
Step 2: Match with options
The option "synthesis and combustion" fits as it is a synthesis (combination) and also a combustion (reaction with $\ce{O_{2}}$ to burn S).
For the reaction $\boldsymbol{\ce{CaCl_{2} + 2AgNO_{3}
ightarrow ...}}$ (assuming it forms $\ce{AgCl}$ precipitate, a double - displacement reaction, but since we need to pick from given options, likely a typo, but if we consider the options, none of the given options (synthesis, decomposition, combustion) fit double - displacement. However, if we assume a different context, but based on the options, maybe a mistake. But focusing on the first reaction:
Step 1: Recall decomposition
Decomposition: A compound breaks into simpler substances. $\ce{2Na_{2}O}$ → $\ce{4Na + O_{2}}$ is decomposition (one compound → two elements).
Step 2: Match with options
The option "decomposition only" fits.
For $\boldsymbol{\ce{Zn + CuSO_{4}
ightarrow ZnSO_{4} + Cu}}$ (single - displacement: A + BC → AC + B, not matching synthesis, decomposition, or combustion options. But if we follow the options, maybe a mistake. However, focusing on the given reactions with clear matches:
For $\ce{S + O_{2}
ightarrow SO_{2}}$: synthesis and combustion
For $\ce{2Na_{2}O
ightarrow 4Na + O_{2}}$: decomposition only
(Note: The $\ce{CaCl_{2} + 2AgNO_{3}}$ and $\ce{Zn + CuSO_{4}}$ reactions do not fit the given option types (synthesis, decomposition, combustion) as they are double - displacement and single - displacement respectively, suggesting a possible error in the problem setup, but the key reactions with clear matches are analyzed above.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
for $\boldsymbol{\ce{S + O_{2}
ightarrow SO_{2}}}$:
synthesis and combustion
For $\boldsymbol{\ce{2Na_{2}O
ightarrow 4Na + O_{2}}}$ (decomposition: AB → A + B, here $\ce{Na_{2}O}$ breaks into $\ce{Na}$ and $\ce{O_{2}}$):