QUESTION IMAGE
Question
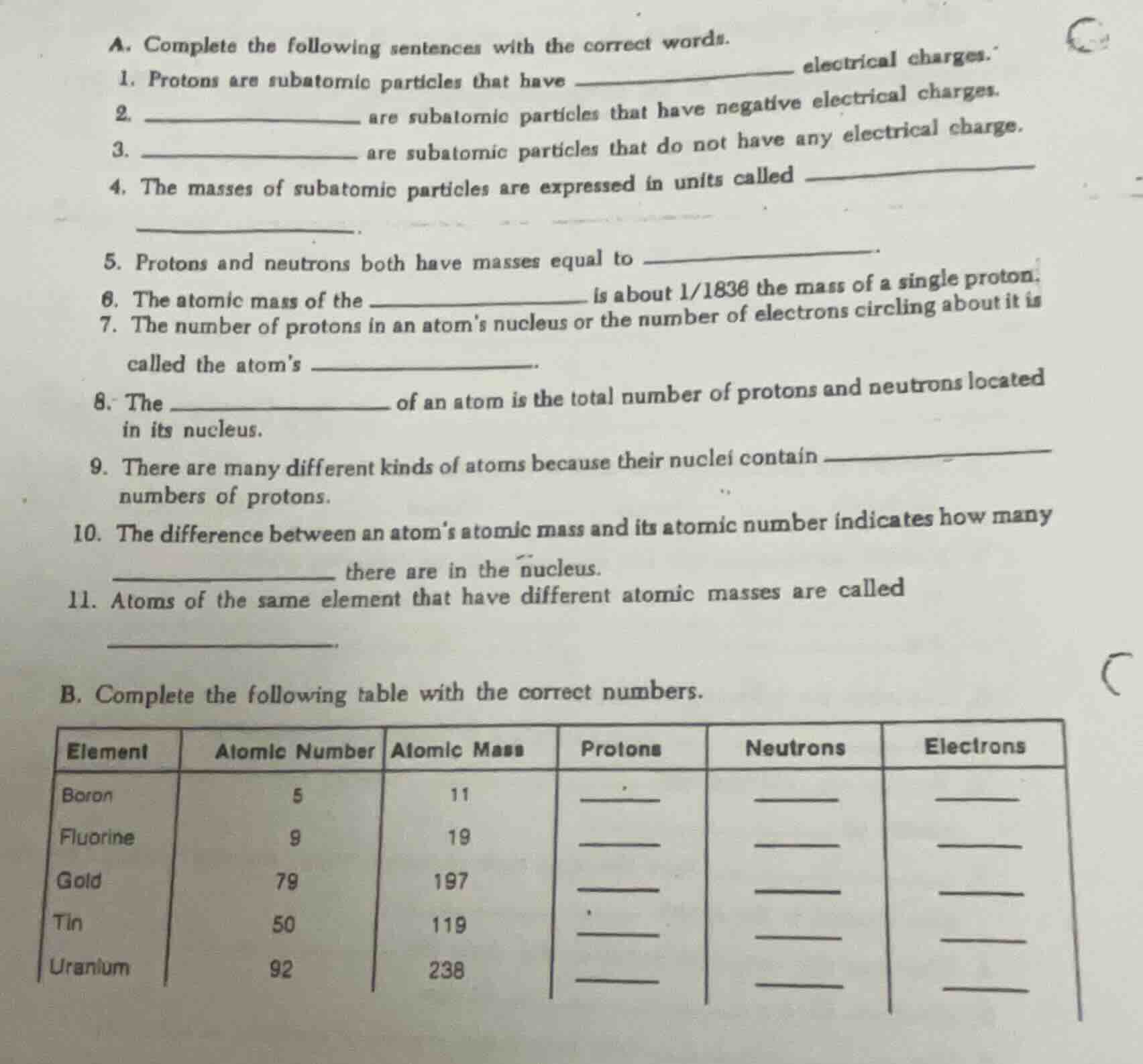
a. complete the following sentences with the correct words.
- protons are subatomic particles that have ______ electrical charges.
- ______ are subatomic particles that have negative electrical charges.
- ______ are subatomic particles that do not have any electrical charge.
- the masses of subatomic particles are expressed in units called ______.
- protons and neutrons both have masses equal to ______.
- the atomic mass of the ______ is about 1/1836 the mass of a single proton.
- the number of protons in an atoms nucleus or the number of electrons circling about it is called the atoms ______.
- the ______ of an atom is the total number of protons and neutrons located in its nucleus.
- there are many different kinds of atoms because their nuclei contain ______ numbers of protons.
- the difference between an atoms atomic mass and its atomic number indicates how many ______ there are in the nucleus.
- atoms of the same element that have different atomic masses are called ______.
b. complete the following table with the correct numbers.
| element | atomic number | atomic mass | protons | neutrons | electrons |
|---|---|---|---|---|---|
| fluorine | 9 | 19 | ______ | ______ | ______ |
| gold | 79 | 197 | ______ | ______ | ______ |
| tin | 50 | 119 | ______ | ______ | ______ |
| uranium | 92 | 238 | ______ | ______ | ______ |
Part A:
Step1: Recall proton charge property
Protons carry positive charge.
Step2: Recall negative subatomic particle
Electrons have negative electrical charge.
Step3: Recall neutral subatomic particle
Neutrons have no electrical charge.
Step4: Recall subatomic mass unit
Subatomic masses use atomic mass units.
Step5: Recall proton/neutron mass value
Protons/neutrons = 1 amu each.
Step6: Recall electron mass relative to proton
Electron mass ≈ 1/1836 of proton.
Step7: Recall proton/electron count term
Proton/electron count = atomic number.
Step8: Recall proton+neutron count term
Proton+neutron sum = atomic mass.
Step9: Recall proton count & atom identity
Different proton counts = different atoms.
Step10: Calculate neutron count formula
Neutrons = Atomic mass - Atomic number.
Step11: Recall same-element mass variants
Same element, different masses = isotopes.
Part B:
Step1: Protons = Atomic Number
Protons = Atomic Number, so copy atomic number value.
Step2: Electrons = Protons (neutral atom)
Electrons = Protons for neutral atoms, copy proton value.
Step3: Neutrons = Atomic Mass - Protons
Neutrons = Atomic Mass - Protons, calculate:
- Boron: $11 - 5 = 6$
- Fluorine: $19 - 9 = 10$
- Gold: $197 - 79 = 118$
- Tin: $119 - 50 = 69$
- Uranium: $238 - 92 = 146$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part A
- positive
- Electrons
- Neutrons
- atomic mass units (amu)
- 1 atomic mass unit (amu)
- electron
- atomic number
- atomic mass
- different
- neutrons
- isotopes
Part B
| Element | Atomic Number | Atomic Mass | Protons | Neutrons | Electrons |
|---|---|---|---|---|---|
| Fluorine | 9 | 19 | 9 | 10 | 9 |
| Gold | 79 | 197 | 79 | 118 | 79 |
| Tin | 50 | 119 | 50 | 69 | 50 |
| Uranium | 92 | 238 | 92 | 146 | 92 |