QUESTION IMAGE
Question
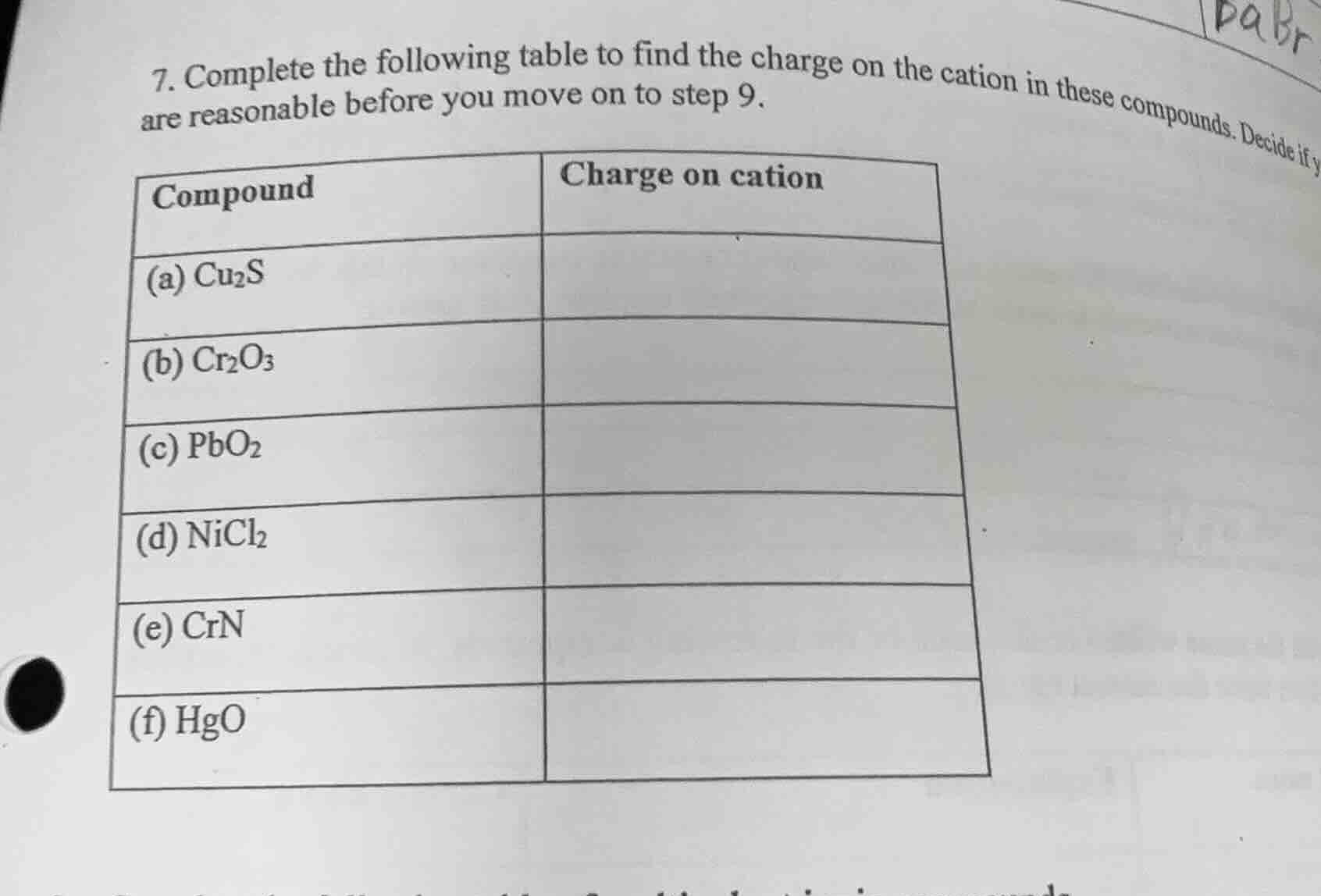
- complete the following table to find the charge on the cation in these compounds. decide if y are reasonable before you move on to step 9.
| compound | charge on cation |
|---|---|
| (b) $\ce{cr_{2}o_{3}}$ | |
| (c) $\ce{pbo_{2}}$ | |
| (d) $\ce{nicl_{2}}$ | |
| (e) $\ce{crn}$ | |
| (f) $\ce{hgo}$ |
Step1: Recall anion charges
Sulfide ($\text{S}^{2-}$), oxide ($\text{O}^{2-}$), chloride ($\text{Cl}^-$), nitride ($\text{N}^{3-}$) are standard anions with fixed charges.
Step2: Neutrality rule for (a) $\text{Cu}_2\text{S}$
Total charge = 0. Let $\text{Cu}$ charge = $x$.
$$2x + (-2) = 0 \implies x = +1$$
Step3: Neutrality rule for (b) $\text{Cr}_2\text{O}_3$
Let $\text{Cr}$ charge = $x$.
$$2x + 3(-2) = 0 \implies 2x = 6 \implies x = +3$$
Step4: Neutrality rule for (c) $\text{PbO}_2$
Let $\text{Pb}$ charge = $x$.
$$x + 2(-2) = 0 \implies x = +4$$
Step5: Neutrality rule for (d) $\text{NiCl}_2$
Let $\text{Ni}$ charge = $x$.
$$x + 2(-1) = 0 \implies x = +2$$
Step6: Neutrality rule for (e) $\text{CrN}$
Let $\text{Cr}$ charge = $x$.
$$x + (-3) = 0 \implies x = +3$$
Step7: Neutrality rule for (f) $\text{HgO}$
Let $\text{Hg}$ charge = $x$.
$$x + (-2) = 0 \implies x = +2$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Compound | Charge on cation |
|---|---|
| (b) $\text{Cr}_2\text{O}_3$ | +3 |
| (c) $\text{PbO}_2$ | +4 |
| (d) $\text{NiCl}_2$ | +2 |
| (e) $\text{CrN}$ | +3 |
| (f) $\text{HgO}$ | +2 |