QUESTION IMAGE
Question
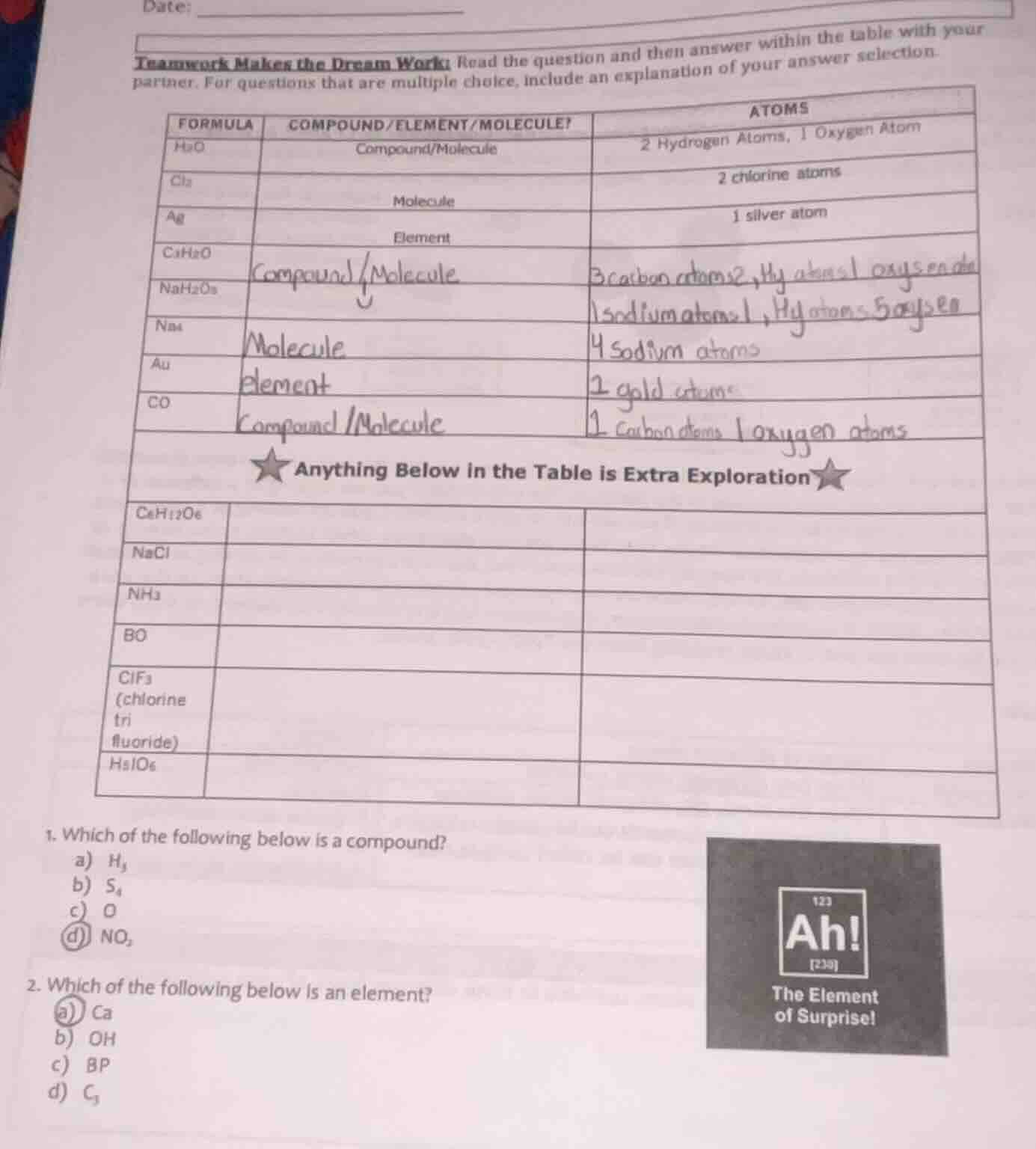
date:
teamwork makes the dream work: read the question and then answer within the table with your partner. for questions that are multiple choice, include an explanation of your answer selection.
| formula | compound/element/molecule? | atoms |
|---|---|---|
| cl₂ | molecule | 2 chlorine atoms |
| ag | element | 1 silver atom |
| c₃h₆o | ||
| nah₂o₅ | ||
| na₄ | ||
| au | ||
| co | ||
| ★ anything below in the table is extra exploration ★ | ||
| c₆h₁₂o₆ | ||
| nacl | ||
| nh₃ | ||
| bo | ||
| clf₃ (chlorine tri fluoride) | ||
| h₅io₆ |
- which of the following below is a compound?
a) h₂
b) s₄
c) o
d) no₂
- which of the following below is an element?
a) ca
b) oh
c) bp
d) c₃
image: a square with 123 ah! 239 the element of surprise!
Question 1
A compound is a substance made of two or more different elements chemically bonded. \(H_2\) (a) and \(S_4\) (b) are molecules of single elements (hydrogen, sulfur). \(O\) (c) is an element. \(NO_2\) (d) has nitrogen and oxygen (two different elements), so it's a compound.
An element is a pure substance with one type of atom. \(Ca\) (a) is a single element (calcium). \(OH\) (b) is a group (not an element), \(BP\) (c) has two elements (boron, phosphorus), \(C_3\) (d) is a molecule of carbon (but the question likely expects the single - atom element representation; \(Ca\) is a single element symbol).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. \(NO_2\)