QUESTION IMAGE
Question
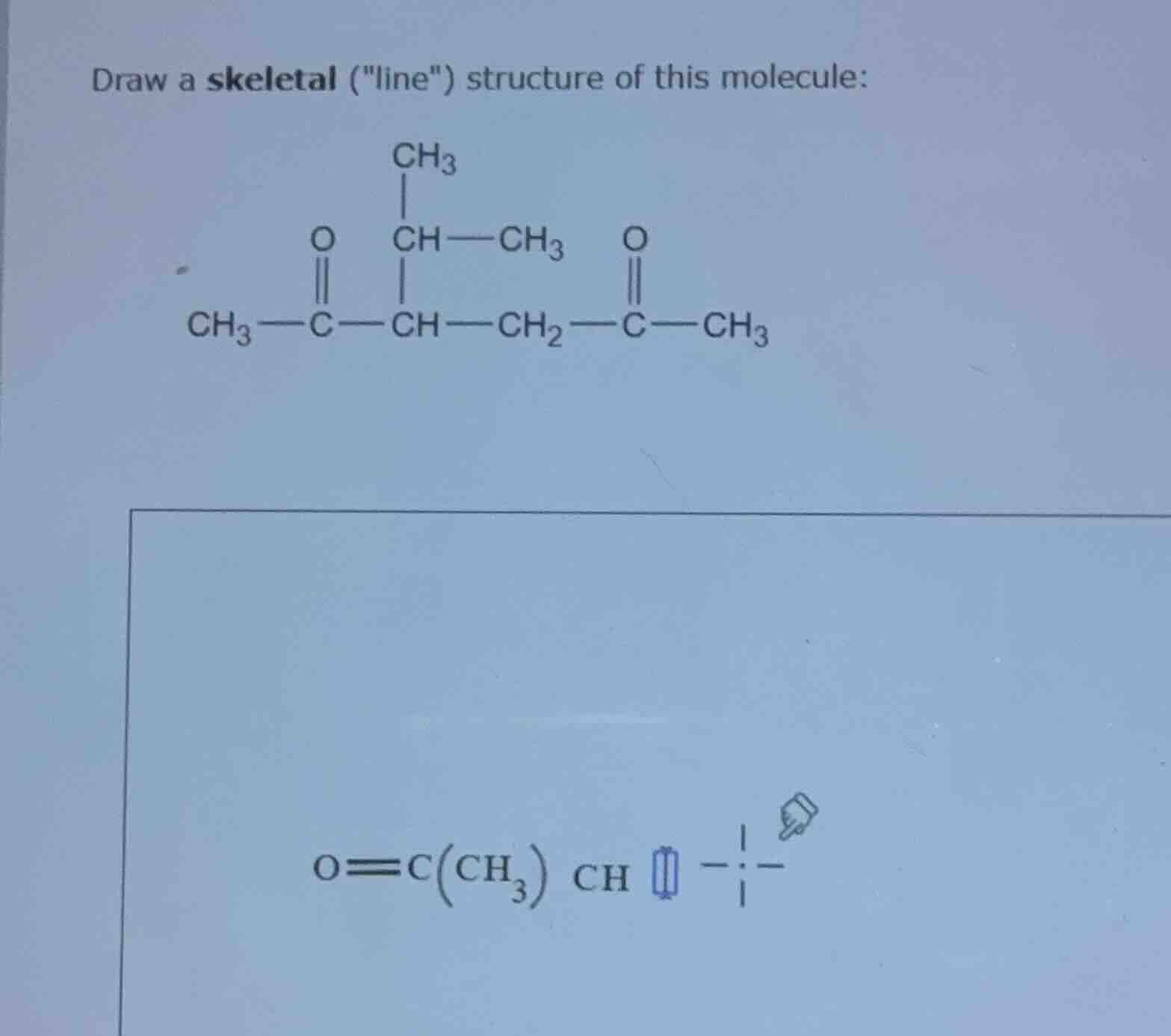
draw a skeletal (\line\) structure of this molecule:
$\ce{ch3 - c(=o) - ch(ch(ch3)ch3) - ch2 - c(=o) - ch3}$
Step1: Identify Carbon Atoms and Functional Groups
First, we analyze the given molecular formula. The molecule has carbonyl groups (\(C=O\)) and various carbon - carbon bonds. Let's list the carbon atoms:
- The first carbonyl carbon: \(C\) in \(CH_3 - C(=O)-\)
- Then the carbon chain: \( - CH - CH_2 - C(=O)-CH_3\), and there is a branch at the second carbon (from the left carbonyl) with \(CH - CH_3\) and \(CH_3\) attached.
Step2: Draw the Skeletal Structure Basics
In skeletal (line) structure, we represent carbon atoms as vertices (or the ends of lines). We start by drawing the main carbon chain.
- The main chain has carbon atoms: from the first carbonyl (\(C = O\)) carbon, then a \(CH\) (with a branch), then a \(CH_2\), then a carbonyl \(C = O\), then a \(CH_3\).
- The branch at the second carbon (after the first carbonyl) has a \(CH\) (attached to \(CH_3\)) and another \(CH_3\) attached to that \(CH\).
Step3: Add Functional Groups and Branches
- Draw the two carbonyl groups (\(C = O\)) as double - bonded \(O\) to the appropriate carbon atoms.
- For the branch: at the carbon between the two carbonyl groups (the second carbon in the main chain, counting from the left carbonyl), we attach a branch with two \(CH_3\) groups (one directly on the \(CH\) and one on the adjacent \(CH\) in the branch).
The skeletal structure should look like this:
First, draw the main chain:
\(O=\) (attached to a carbon), then a line (representing \(CH\)) with a branch. The branch has a line (representing \(CH\)) with a \(CH_3\) at the end and another \(CH_3\) on the \(CH\) of the branch. Then a \(CH_2\) line, then \(O=\) (attached to a carbon), then a \(CH_3\).
In more precise terms, the skeletal structure is:
CH3
|
O | O
|| | ||
C - C - C - C - C - CH3
| |
CH3 CH2Wait, let's re - elaborate. Let's number the carbons:
- Carbon 1: \(C = O\) (attached to \(CH_3\))
- Carbon 2: \(CH\) (with a branch)
- Carbon 3: \(CH_2\)
- Carbon 4: \(C = O\) (attached to \(CH_3\))
Branch on Carbon 2: Carbon 2 is bonded to a \(CH\) (Carbon 2a) which is bonded to \(CH_3\) (Carbon 2b) and another \(CH_3\) (on Carbon 2a).
So the skeletal structure:
CH3
|
O | O
|| | ||
C - C - C - C - CH3
|
CH - CH3
|
CH3Wait, no, let's correct the carbon numbering. Let's start from the left - most carbon:
- Left - most carbon: \(C\) in \(CH_3 - C(=O)-\) (Carbon 1)
- Carbon 2: \(CH\) (between Carbon 1 and Carbon 3)
- Carbon 3: \(CH_2\)
- Carbon 4: \(C(=O)\) (in \( - C(=O)-CH_3\))
- Carbon 5: \(CH_3\) (attached to Carbon 4)
Branch on Carbon 2: Carbon 2 is bonded to a \(CH\) (Carbon 6) which is bonded to \(CH_3\) (Carbon 7) and \(CH_3\) (Carbon 8) is bonded to Carbon 6.
So the skeletal structure:
CH3 (Carbon 8)
|
C (Carbon 6) - CH3 (Carbon 7)
|
O | O
|| | ||
C (1) - C (2) - C (3) - C (4) - CH3 (5)In a more standard skeletal drawing:
CH3
|
O | O
|| | ||
C - C - C - C - CH3
|
C - CH3
|
CH3Wait, actually, the correct skeletal structure is:
We can represent it as:
CH3
|
O | O
|| | ||
C - C - C - C - CH3
|
CH - CH3
|
CH3But to draw it properly, the key is to show the carbonyl groups, the main carbon chain, and the branch with two methyl groups.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The skeletal (line) structure of the molecule is:
CH₃
|
O | O
║ | ║
C - C - C - C - CH₃
|
CH - CH₃
|
CH₃(Or in a more compact and standard drawing:
CH3
|
O | O
|| | ||
C - C - C - C - CH3
|
C - CH3
|
CH3where the double lines (\(||\)) represent the carbon - oxygen double bond, and the lines represent carbon - carbon bonds, with the \(CH_3\) groups as branches.)