QUESTION IMAGE
Question
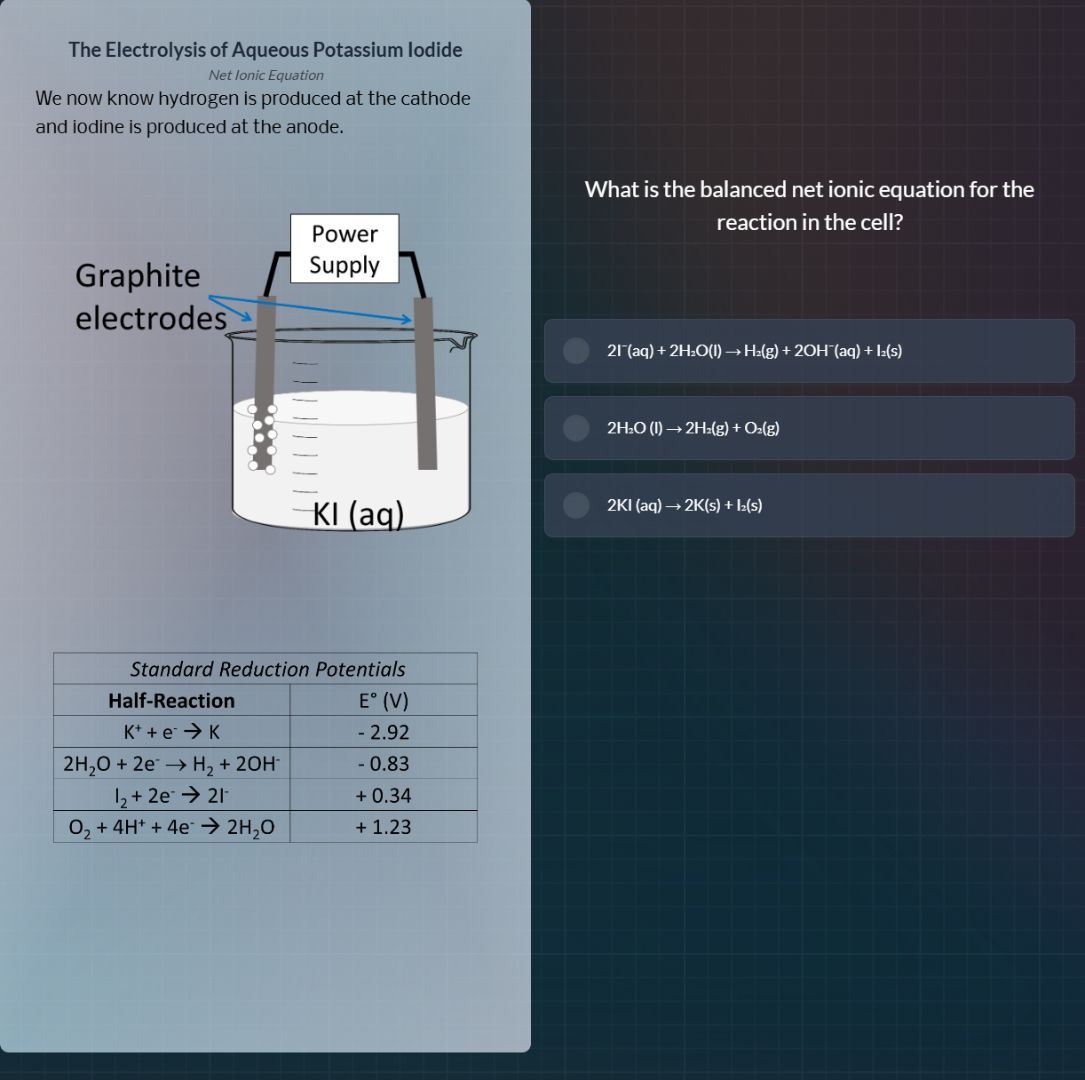
the electrolysis of aqueous potassium iodide
net ionic equation
we now know hydrogen is produced at the cathode
and iodine is produced at the anode.
what is the balanced net ionic equation for the
reaction in the cell?
2i⁻(aq) + 2h₂o(l) → h₂(g) + 2oh⁻(aq) + i₂(s)
2h₂o (l) → 2h₂(g) + o₂(g)
2ki (aq) → 2k(s) + i₂(s)
standard reduction potentials
half - reaction | e° (v)
k⁺ + e⁻ → k | - 2.92
2h₂o + 2e⁻ → h₂ + 2oh⁻ | - 0.83
i₂ + 2e⁻ → 2i⁻ | + 0.34
o₂ + 4h⁺ + 4e⁻ → 2h₂o | + 1.23
Step1: Analyze Cathode (Reduction)
The cathode reaction: We compare reduction potentials. For \( K^+ + e^- \to K \) (\( E^\circ = -2.92 \, V \)) and \( 2H_2O + 2e^- \to H_2 + 2OH^- \) (\( E^\circ = -0.83 \, V \)). The less negative (more positive) potential means \( 2H_2O + 2e^- \to H_2 + 2OH^- \) occurs (reduction at cathode).
Step2: Analyze Anode (Oxidation)
The anode reaction (oxidation is reverse of reduction). Possible oxidations: reverse of \( I_2 + 2e^- \to 2I^- \) (\( E^\circ = +0.34 \, V \)) gives \( 2I^- \to I_2 + 2e^- \) (oxidation, since it's reverse of reduction with lower \( E^\circ \) than reverse of \( O_2 + 4H^+ + 4e^- \to 2H_2O \) (\( E^\circ = +1.23 \, V \))).
Step3: Balance Electrons and Combine Reactions
Cathode: \( 2H_2O + 2e^- \to H_2 + 2OH^- \) (2 electrons)
Anode: \( 2I^- \to I_2 + 2e^- \) (2 electrons)
Add them: \( 2I^-(aq) + 2H_2O(l) \to H_2(g) + 2OH^-(aq) + I_2(s) \)
Check other options:
- \( 2H_2O(l) \to 2H_2(g) + O_2(g) \): Not matching (no \( I_2 \) or \( OH^- \), \( I^- \))
- \( 2KI(aq) \to 2K(s) + I_2(s) \): \( K^+ \) reduction is not favorable (low \( E^\circ \)), so K doesn't form.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 2I^-(aq) + 2H_2O(l) \to H_2(g) + 2OH^-(aq) + I_2(s) \) (the first option)