QUESTION IMAGE
Question
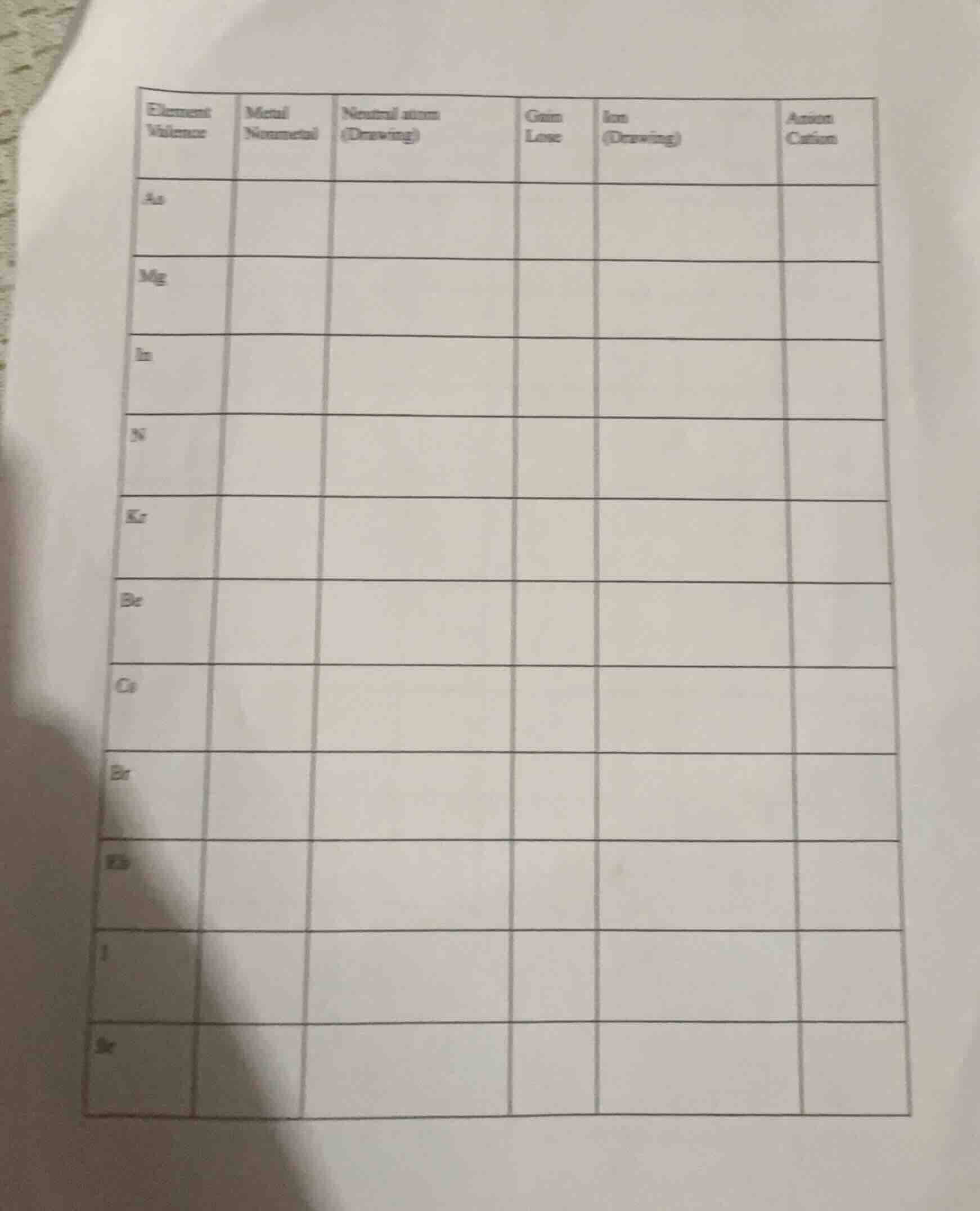
element
valence\tmetal
nonmetal\tneutral atom
(drawing)\tgain
lose\tion
(drawing)\tanion
cation
as\t\t\t\t\t
mg\t\t\t\t\t
in\t\t\t\t\t
n\t\t\t\t\t
kr\t\t\t\t\t
be\t\t\t\t\t
ca\t\t\t\t\t
br\t\t\t\t\t
k\t\t\t\t\t
i\t\t\t\t\t
sr\t\t\t\t\t
To solve this problem, we analyze each element based on its properties (metal/nonmetal, valence, electron gain/loss, ion type, and Lewis dot structures for neutral atoms and ions). Here's the breakdown for each element:
1. Arsenic (As)
- Metal/Nonmetal: Nonmetal (metalloid, but behaves as nonmetal in ion formation).
- Valence: Group 15, valence electrons = 5.
- Neutral Atom (Lewis Dot): 5 valence electrons (e.g., $\dot{\text{As}}\dot{}$ with 3 lone pairs and 2 unpaired, or standard Lewis dot: $\text{As:}$ with 3 lone pairs and 2 bonding electrons, but typically drawn as 5 dots around As).
- Gain/Lose Electrons: Gains 3 electrons (to achieve octet, $8 - 5 = 3$).
- Ion (Lewis Dot): $\text{As}^{3-}$ (8 valence electrons, 3 extra; Lewis dot: $[:\text{As}:]^{3-}$).
- Anion/Cation: Anion (gains electrons).
2. Magnesium (Mg)
- Metal/Nonmetal: Metal.
- Valence: Group 2, valence electrons = 2.
- Neutral Atom (Lewis Dot): 2 valence electrons (e.g., $\text{Mg}\cdot\cdot$).
- Gain/Lose Electrons: Loses 2 electrons (to achieve octet, $2 - 2 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{Mg}^{2+}$ (no valence electrons; Lewis dot: $\text{Mg}^{2+}$).
- Anion/Cation: Cation (loses electrons).
3. Indium (In)
- Metal/Nonmetal: Metal (Group 13, post-transition metal).
- Valence: Group 13, valence electrons = 3.
- Neutral Atom (Lewis Dot): 3 valence electrons (e.g., $\dot{\text{In}}\dot{}\dot{}$).
- Gain/Lose Electrons: Loses 3 electrons (to achieve octet, $3 - 3 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{In}^{3+}$ (no valence electrons; Lewis dot: $\text{In}^{3+}$).
- Anion/Cation: Cation (loses electrons).
4. Nitrogen (N)
- Metal/Nonmetal: Nonmetal.
- Valence: Group 15, valence electrons = 5.
- Neutral Atom (Lewis Dot): 5 valence electrons (e.g., $\text{N:}$ with 3 lone pairs and 2 bonding electrons, or $\dot{\text{N}}\dot{}$ with 3 lone pairs and 2 unpaired).
- Gain/Lose Electrons: Gains 3 electrons (to achieve octet, $8 - 5 = 3$).
- Ion (Lewis Dot): $\text{N}^{3-}$ (8 valence electrons; Lewis dot: $[:\text{N}:]^{3-}$).
- Anion/Cation: Anion (gains electrons).
5. Krypton (Kr)
- Metal/Nonmetal: Nonmetal (noble gas).
- Valence: Group 18, valence electrons = 8 (stable octet).
- Neutral Atom (Lewis Dot): 8 valence electrons (e.g., $[:\text{Kr}:]$).
- Gain/Lose Electrons: Neither (already stable).
- Ion (Lewis Dot): No ion (stable as neutral atom).
- Anion/Cation: Neither (noble gas, no ion formation).
6. Beryllium (Be)
- Metal/Nonmetal: Metal.
- Valence: Group 2, valence electrons = 2.
- Neutral Atom (Lewis Dot): 2 valence electrons (e.g., $\text{Be}\cdot\cdot$).
- Gain/Lose Electrons: Loses 2 electrons (to achieve octet, $2 - 2 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{Be}^{2+}$ (no valence electrons; Lewis dot: $\text{Be}^{2+}$).
- Anion/Cation: Cation (loses electrons).
7. Calcium (Ca)
- Metal/Nonmetal: Metal.
- Valence: Group 2, valence electrons = 2.
- Neutral Atom (Lewis Dot): 2 valence electrons (e.g., $\text{Ca}\cdot\cdot$).
- Gain/Lose Electrons: Loses 2 electrons (to achieve octet, $2 - 2 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{Ca}^{2+}$ (no valence electrons; Lewis dot: $\text{Ca}^{2+}$).
- Anion/Cation: Cation (loses electrons).
8. Bromine (Br)
- Metal/Nonmetal: Nonmetal.
- Valence: Group 17, valence electrons = 7.
- *Neutral Atom (Lewis Dot)…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this problem, we analyze each element based on its properties (metal/nonmetal, valence, electron gain/loss, ion type, and Lewis dot structures for neutral atoms and ions). Here's the breakdown for each element:
1. Arsenic (As)
- Metal/Nonmetal: Nonmetal (metalloid, but behaves as nonmetal in ion formation).
- Valence: Group 15, valence electrons = 5.
- Neutral Atom (Lewis Dot): 5 valence electrons (e.g., $\dot{\text{As}}\dot{}$ with 3 lone pairs and 2 unpaired, or standard Lewis dot: $\text{As:}$ with 3 lone pairs and 2 bonding electrons, but typically drawn as 5 dots around As).
- Gain/Lose Electrons: Gains 3 electrons (to achieve octet, $8 - 5 = 3$).
- Ion (Lewis Dot): $\text{As}^{3-}$ (8 valence electrons, 3 extra; Lewis dot: $[:\text{As}:]^{3-}$).
- Anion/Cation: Anion (gains electrons).
2. Magnesium (Mg)
- Metal/Nonmetal: Metal.
- Valence: Group 2, valence electrons = 2.
- Neutral Atom (Lewis Dot): 2 valence electrons (e.g., $\text{Mg}\cdot\cdot$).
- Gain/Lose Electrons: Loses 2 electrons (to achieve octet, $2 - 2 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{Mg}^{2+}$ (no valence electrons; Lewis dot: $\text{Mg}^{2+}$).
- Anion/Cation: Cation (loses electrons).
3. Indium (In)
- Metal/Nonmetal: Metal (Group 13, post-transition metal).
- Valence: Group 13, valence electrons = 3.
- Neutral Atom (Lewis Dot): 3 valence electrons (e.g., $\dot{\text{In}}\dot{}\dot{}$).
- Gain/Lose Electrons: Loses 3 electrons (to achieve octet, $3 - 3 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{In}^{3+}$ (no valence electrons; Lewis dot: $\text{In}^{3+}$).
- Anion/Cation: Cation (loses electrons).
4. Nitrogen (N)
- Metal/Nonmetal: Nonmetal.
- Valence: Group 15, valence electrons = 5.
- Neutral Atom (Lewis Dot): 5 valence electrons (e.g., $\text{N:}$ with 3 lone pairs and 2 bonding electrons, or $\dot{\text{N}}\dot{}$ with 3 lone pairs and 2 unpaired).
- Gain/Lose Electrons: Gains 3 electrons (to achieve octet, $8 - 5 = 3$).
- Ion (Lewis Dot): $\text{N}^{3-}$ (8 valence electrons; Lewis dot: $[:\text{N}:]^{3-}$).
- Anion/Cation: Anion (gains electrons).
5. Krypton (Kr)
- Metal/Nonmetal: Nonmetal (noble gas).
- Valence: Group 18, valence electrons = 8 (stable octet).
- Neutral Atom (Lewis Dot): 8 valence electrons (e.g., $[:\text{Kr}:]$).
- Gain/Lose Electrons: Neither (already stable).
- Ion (Lewis Dot): No ion (stable as neutral atom).
- Anion/Cation: Neither (noble gas, no ion formation).
6. Beryllium (Be)
- Metal/Nonmetal: Metal.
- Valence: Group 2, valence electrons = 2.
- Neutral Atom (Lewis Dot): 2 valence electrons (e.g., $\text{Be}\cdot\cdot$).
- Gain/Lose Electrons: Loses 2 electrons (to achieve octet, $2 - 2 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{Be}^{2+}$ (no valence electrons; Lewis dot: $\text{Be}^{2+}$).
- Anion/Cation: Cation (loses electrons).
7. Calcium (Ca)
- Metal/Nonmetal: Metal.
- Valence: Group 2, valence electrons = 2.
- Neutral Atom (Lewis Dot): 2 valence electrons (e.g., $\text{Ca}\cdot\cdot$).
- Gain/Lose Electrons: Loses 2 electrons (to achieve octet, $2 - 2 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{Ca}^{2+}$ (no valence electrons; Lewis dot: $\text{Ca}^{2+}$).
- Anion/Cation: Cation (loses electrons).
8. Bromine (Br)
- Metal/Nonmetal: Nonmetal.
- Valence: Group 17, valence electrons = 7.
- Neutral Atom (Lewis Dot): 7 valence electrons (e.g., $\dot{\text{Br}}\dot{}\dot{}\dot{}\dot{}$ with 3 lone pairs and 1 unpaired).
- Gain/Lose Electrons: Gains 1 electron (to achieve octet, $8 - 7 = 1$).
- Ion (Lewis Dot): $\text{Br}^{-}$ (8 valence electrons; Lewis dot: $[:\text{Br}:]^{-}$).
- Anion/Cation: Anion (gains electrons).
9. Potassium (K)
- Metal/Nonmetal: Metal.
- Valence: Group 1, valence electrons = 1.
- Neutral Atom (Lewis Dot): 1 valence electron (e.g., $\text{K}\cdot$).
- Gain/Lose Electrons: Loses 1 electron (to achieve octet, $1 - 1 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{K}^{+}$ (no valence electrons; Lewis dot: $\text{K}^{+}$).
- Anion/Cation: Cation (loses electrons).
10. Iodine (I)
- Metal/Nonmetal: Nonmetal.
- Valence: Group 17, valence electrons = 7.
- Neutral Atom (Lewis Dot): 7 valence electrons (e.g., $\dot{\text{I}}\dot{}\dot{}\dot{}\dot{}$ with 3 lone pairs and 1 unpaired).
- Gain/Lose Electrons: Gains 1 electron (to achieve octet, $8 - 7 = 1$).
- Ion (Lewis Dot): $\text{I}^{-}$ (8 valence electrons; Lewis dot: $[:\text{I}:]^{-}$).
- Anion/Cation: Anion (gains electrons).
11. Strontium (Sr)
- Metal/Nonmetal: Metal.
- Valence: Group 2, valence electrons = 2.
- Neutral Atom (Lewis Dot): 2 valence electrons (e.g., $\text{Sr}\cdot\cdot$).
- Gain/Lose Electrons: Loses 2 electrons (to achieve octet, $2 - 2 = 0$ for octet in next shell).
- Ion (Lewis Dot): $\text{Sr}^{2+}$ (no valence electrons; Lewis dot: $\text{Sr}^{2+}$).
- Anion/Cation: Cation (loses electrons).
Filled Table (Summary)
| Element | Metal/Nonmetal | Valence | Neutral Atom (Lewis Dot) | Gain/Lose Electrons | Ion (Lewis Dot) | Anion/Cation |
|---|---|---|---|---|---|---|
| Mg | Metal | 2 | $\text{Mg}\cdot\cdot$ | Loses 2 | $\text{Mg}^{2+}$ | Cation |
| In | Metal | 3 | $\dot{\text{In}}\dot{}\dot{}$ | Loses 3 | $\text{In}^{3+}$ | Cation |
| N | Nonmetal | 5 | $\text{N:}$ (5 dots) | Gains 3 | $[:\text{N}:]^{3-}$ | Anion |
| Kr | Nonmetal | 8 | $[:\text{Kr}:]$ | Neither | None | Neither |
| Be | Metal | 2 | $\text{Be}\cdot\cdot$ | Loses 2 | $\text{Be}^{2+}$ | Cation |
| Ca | Metal | 2 | $\text{Ca}\cdot\cdot$ | Loses 2 | $\text{Ca}^{2+}$ | Cation |
| Br | Nonmetal | 7 | $\dot{\text{Br}}\dot{}\dot{}\dot{}\dot{}$ | Gains 1 | $[:\text{Br}:]^{-}$ | Anion |
| K | Metal | 1 | $\text{K}\cdot$ | Loses 1 | $\text{K}^{+}$ | Cation |
| I | Nonmetal | 7 | $\dot{\text{I}}\dot{}\dot{}\dot{}\dot{}$ | Gains 1 | $[:\text{I}:]^{-}$ | Anion |
| Sr | Metal | 2 | $\text{Sr}\cdot\cdot$ | Loses 2 | $\text{Sr}^{2+}$ | Cation |
Key Notes:
- Lewis Dots: Represent valence electrons (dots) around the element symbol.
- Electron Gain/Loss: Based on achieving a stable octet (8 electrons, except H/He).
- Ion Type: Cations (metals) lose electrons; Anions (nonmetals) gain electrons.
This table summarizes the properties of each element, including Lewis dot structures for neutral atoms and ions, electron behavior, and ion type.