QUESTION IMAGE
Question
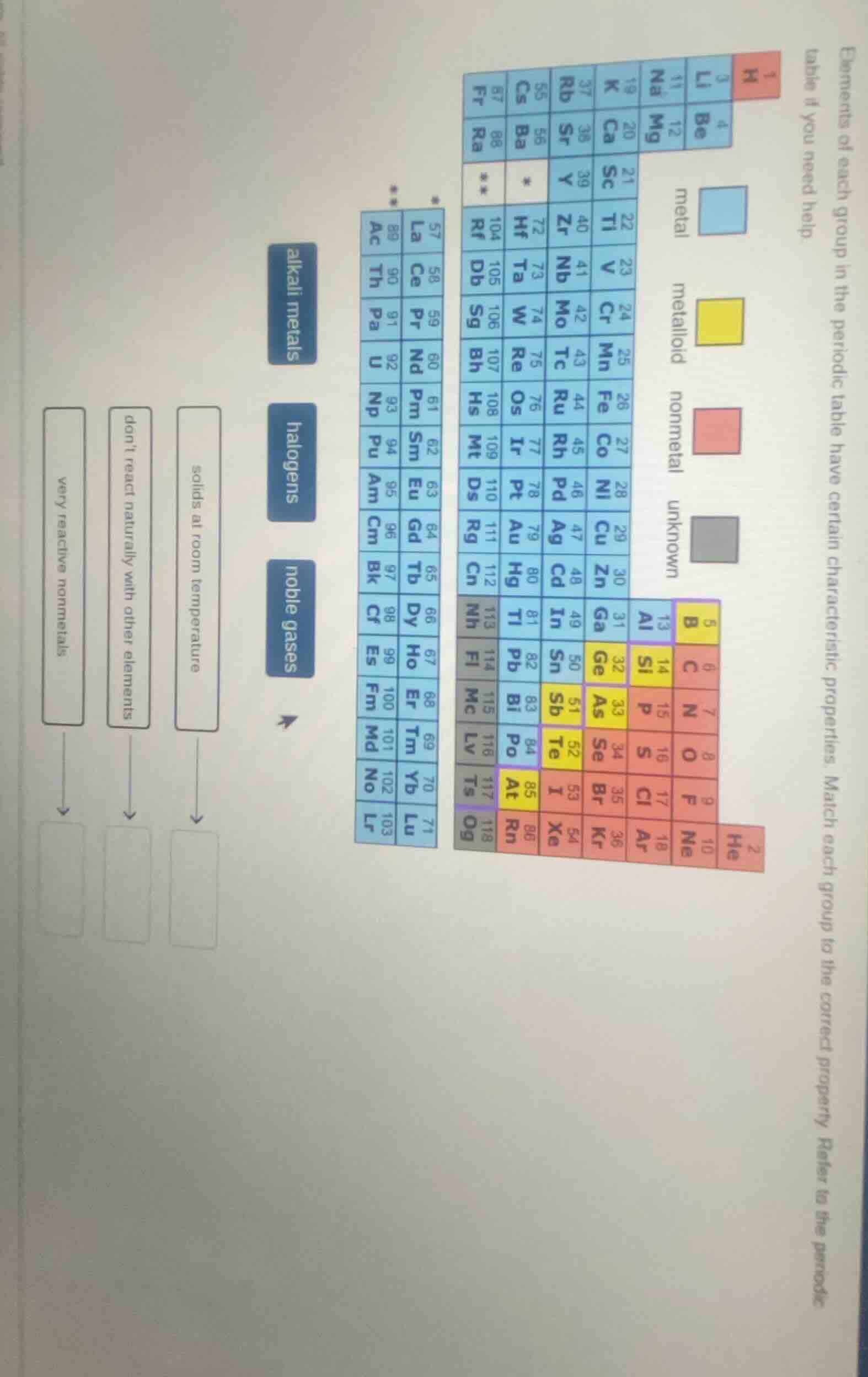
elements of each group in the periodic table have certain characteristic properties. match each group to the correct property. refer to the periodic table if you need help.
(there are boxes with labels: alkali metals, halogens, noble gases on the left, and descriptions very reactive nonmetals, dont react naturally with other elements, solids at room temperature with arrows to empty boxes, along with a periodic table showing metals, metalloids, nonmetals, unknown elements colored differently.)
Para resolver el emparejamiento, analizamos las propiedades de cada grupo:
- Halogens (halógenos): Son no metales muy reactivos (reaccionan fácilmente con metales, por ejemplo). Así que "very reactive nonmetals" se empareja con halogens.
- Noble gases (gases nobles): Son inertes, es decir, "don't react naturally with other elements" (no reaccionan naturalmente con otros elementos).
- Alkali metals (metales alcalinos): La mayoría son sólidos a temperatura ambiente (como Li, Na, K, etc.). Así que "solids at room temperature" se empareja con alkali metals.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- "very reactive nonmetals" → halogens
- "don't react naturally with other elements" → noble gases
- "solids at room temperature" → alkali metals