QUESTION IMAGE
Question
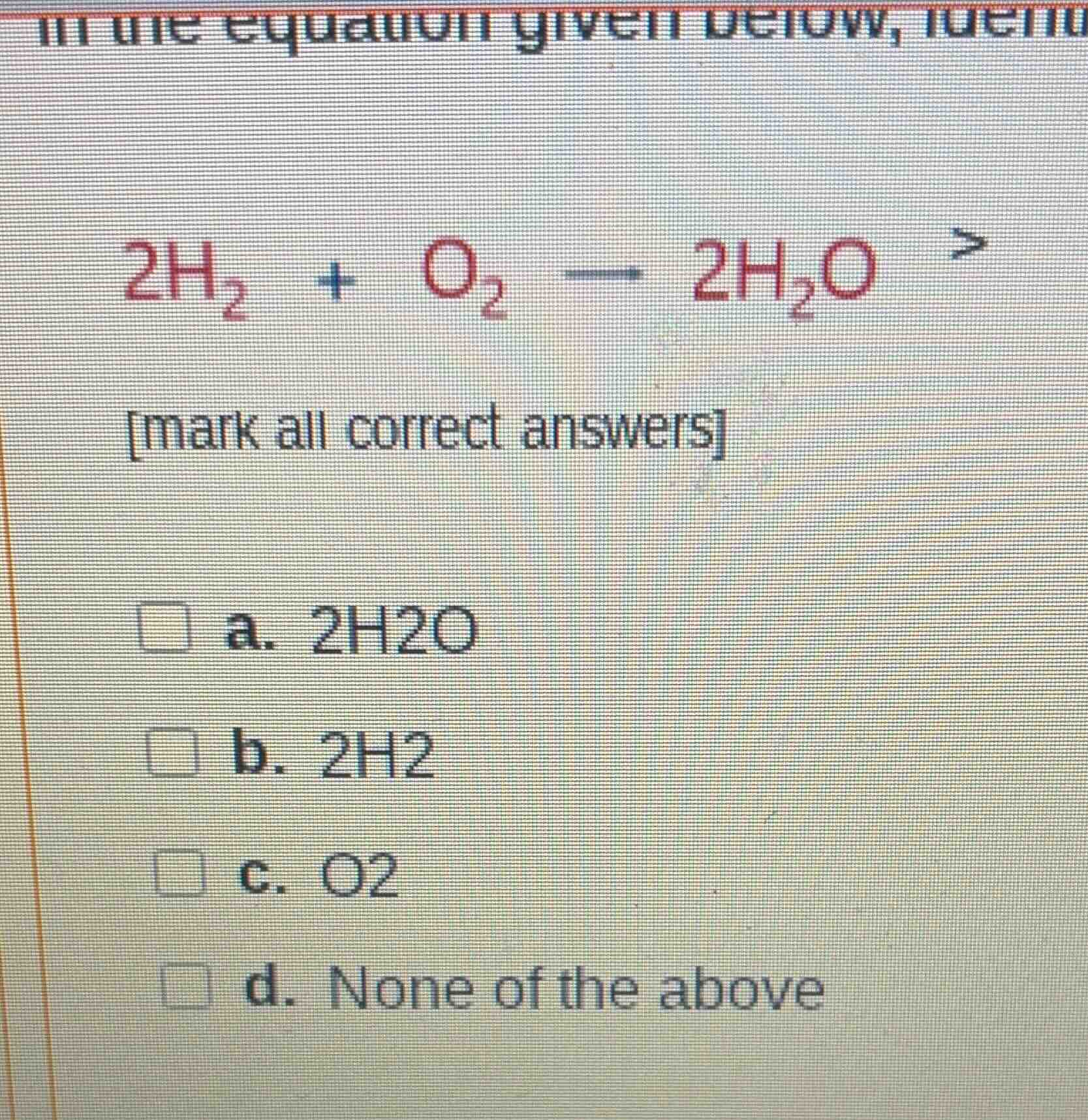
in the equation given below, identify (equation: 2h₂ + o₂ — 2h₂o) mark all correct answers a. 2h2o b. 2h2 c. o2 d. none of the above
Brief Explanations
The chemical equation is \(2H_2 + O_2
ightarrow 2H_2O\). Let's analyze each option:
- Option a: The correct formula for water is \(H_2O\), and the coefficient is 2, so it should be \(2H_2O\) (with subscript 2 for H and O in the formula), not \(2H2O\) (incorrect formatting, likely a typo but the formula representation is wrong here as it should have subscripts as \(H_2O\) not \(H2O\) when written properly, but also the way it's written as \(2H2O\) is non - standard. However, maybe the question is considering the reactants and products. Wait, no, the equation has \(2H_2\) (which can be written as \(2H2\) in a less - standard but maybe acceptable way here), \(O_2\) (written as \(O2\) here), and \(2H_2O\) (written as \(2H2O\) here). So:
- Option b: \(2H2\) represents \(2H_2\) which is a reactant in the equation.
- Option c: \(O2\) represents \(O_2\) which is a reactant in the equation.
- Option a: The product is \(2H_2O\), and \(2H2O\) is a non - standard but maybe intended representation of \(2H_2O\) (assuming the subscript is just not formatted properly). Wait, the equation has \(2H_2O\) as the product. So if we consider the notation in the options (where subscripts are written without the subscript format, like \(H2O\) instead of \(H_2O\)), then \(2H2O\) (option a) is the product, \(2H2\) (option b) is a reactant, and \(O2\) (option c) is a reactant. So all a, b, c are related to the equation. But maybe the question has a typo in the options, but based on the given options:
- Option b: \(2H2\) is \(2H_2\) (hydrogen gas), which is a reactant.
- Option c: \(O2\) is \(O_2\) (oxygen gas), which is a reactant.
- Option a: If we assume that \(2H2O\) is a mis - written \(2H_2O\) (the product), then it is also part of the equation. But maybe the question is considering the correct representations. However, in the context of the equation, \(2H_2\) (b), \(O_2\) (c), and \(2H_2O\) (a, with correct formula intention) are part of the equation. But maybe the options have formatting issues. But according to the given options, b and c are clearly reactants ( \(2H2\) for \(2H_2\) and \(O2\) for \(O_2\) ), and a is the product ( \(2H2O\) for \(2H_2O\) ). So options a, b, c are correct? Wait, but the original equation is \(2H_2+O_2
ightarrow 2H_2O\). So:
- \(2H_2\) (reactant) can be written as \(2H2\) (option b).
- \(O_2\) (reactant) can be written as \(O2\) (option c).
- \(2H_2O\) (product) can be written as \(2H2O\) (option a, assuming the subscript is just not formatted). So all a, b, c are correct. But maybe the question has a different intention. Wait, the options are a. 2H2O, b. 2H2, c. O2, d. None of the above. So based on the equation, \(2H2\) (b) is \(2H_2\) (reactant), \(O2\) (c) is \(O_2\) (reactant), and \(2H2O\) (a) is \(2H_2O\) (product). So a, b, c are correct. But maybe the question has a typo, but as per the given options, b and c are reactants and a is the product (with formatting issues). So we mark b and c, and maybe a. But let's re - check:
The equation is \(2H_2 + O_2
ightarrow 2H_2O\). So:
- Reactants: \(2H_2\) (which is \(2H2\) in option b) and \(O_2\) (which is \(O2\) in option c).
- Product: \(2H_2O\) (which is \(2H2O\) in option a, assuming the subscript is not formatted). So all a, b, c are correct. But maybe the question expects b and c as reactants and a as product, so they are all part of the equation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. 2H2O, b. 2H2, c. O2