QUESTION IMAGE
Question
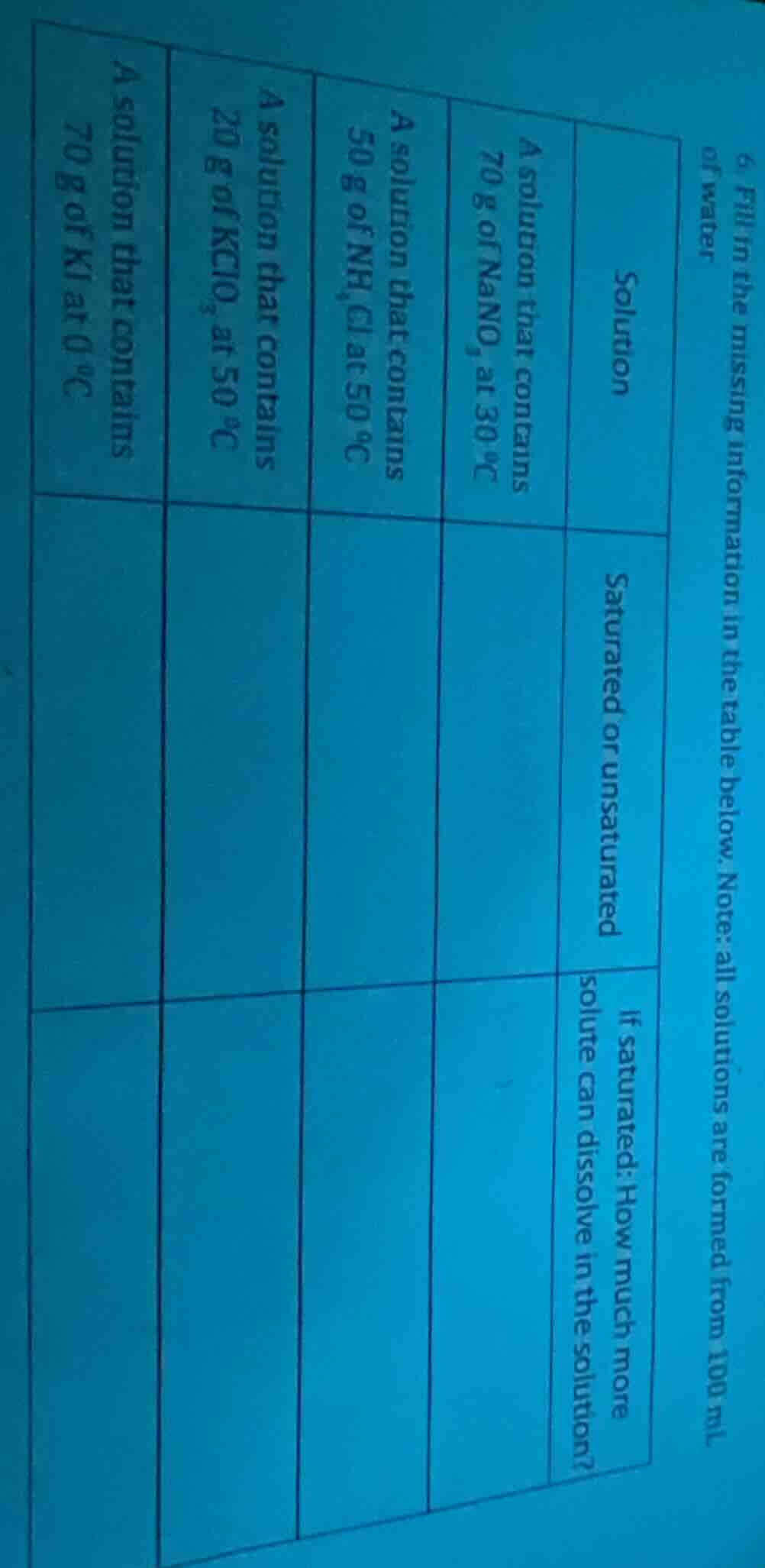
- fill in the missing information in the table below. note: all solutions are formed from 100 ml of water
| solution | saturated or unsaturated | if saturated: how much more solute can dissolve in the solution? |
|---|---|---|
| a solution that contains 50 g of nh₄cl at 50 °c | ||
| a solution that contains 20 g of kclo₃ at 50 °c | ||
| a solution that contains 70 g of ki at 0 °c |
Response
To solve this, we need the solubility values of each solute at the given temperatures (from solubility tables):
1. Solution: A solution that contains 70 g of $\ce{NaNO3}$ at $30^\circ\mathrm{C}$
- Solubility of $\ce{NaNO3}$ at $30^\circ\mathrm{C}$: ~96 g/100 mL water.
- Since $70\ \text{g} < 96\ \text{g}$, the solution is unsaturated.
- No additional solute can dissolve (not saturated).
2. Solution: A solution that contains 50 g of $\ce{NH4Cl}$ at $50^\circ\mathrm{C}$
- Solubility of $\ce{NH4Cl}$ at $50^\circ\mathrm{C}$: ~50 g/100 mL water.
- Since $50\ \text{g} = 50\ \text{g}$, the solution is saturated.
- No more solute can dissolve (already saturated).
3. Solution: A solution that contains 20 g of $\ce{KClO3}$ at $50^\circ\mathrm{C}$
- Solubility of $\ce{KClO3}$ at $50^\circ\mathrm{C}$: ~18 g/100 mL water.
- Since $20\ \text{g} > 18\ \text{g}$, the solution is saturated (excess solute undissolved).
- Additional solute that can dissolve: $18 - 20 = -2$ (but since it’s saturated, no more can dissolve; excess is undissolved). Wait, correction: If solubility is 18 g, then at saturation, only 18 g dissolves. So the solution is saturated (with undissolved solute), and no more can dissolve (already at max, with excess).
4. Solution: A solution that contains 70 g of $\ce{KI}$ at $0^\circ\mathrm{C}$
- Solubility of $\ce{KI}$ at $0^\circ\mathrm{C}$: ~128 g/100 mL water.
- Since $70\ \text{g} < 128\ \text{g}$, the solution is unsaturated.
- No additional solute limit reached (can dissolve more).
Filled Table (Summarized):
| Solution | Saturated or Unsaturated | If Saturated: How much more solute can dissolve? |
|---|---|---|
| $\ce{NH4Cl}$ (50 g, $50^\circ\mathrm{C}$) | Saturated | 0 g (already saturated) |
| $\ce{KClO3}$ (20 g, $50^\circ\mathrm{C}$) | Saturated | 0 g (excess undissolved; max is 18 g) |
| $\ce{KI}$ (70 g, $0^\circ\mathrm{C}$) | Unsaturated | None (unsaturated) |
(Note: Solubility values are approximate from standard solubility curves. Adjust if precise values differ.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we need the solubility values of each solute at the given temperatures (from solubility tables):
1. Solution: A solution that contains 70 g of $\ce{NaNO3}$ at $30^\circ\mathrm{C}$
- Solubility of $\ce{NaNO3}$ at $30^\circ\mathrm{C}$: ~96 g/100 mL water.
- Since $70\ \text{g} < 96\ \text{g}$, the solution is unsaturated.
- No additional solute can dissolve (not saturated).
2. Solution: A solution that contains 50 g of $\ce{NH4Cl}$ at $50^\circ\mathrm{C}$
- Solubility of $\ce{NH4Cl}$ at $50^\circ\mathrm{C}$: ~50 g/100 mL water.
- Since $50\ \text{g} = 50\ \text{g}$, the solution is saturated.
- No more solute can dissolve (already saturated).
3. Solution: A solution that contains 20 g of $\ce{KClO3}$ at $50^\circ\mathrm{C}$
- Solubility of $\ce{KClO3}$ at $50^\circ\mathrm{C}$: ~18 g/100 mL water.
- Since $20\ \text{g} > 18\ \text{g}$, the solution is saturated (excess solute undissolved).
- Additional solute that can dissolve: $18 - 20 = -2$ (but since it’s saturated, no more can dissolve; excess is undissolved). Wait, correction: If solubility is 18 g, then at saturation, only 18 g dissolves. So the solution is saturated (with undissolved solute), and no more can dissolve (already at max, with excess).
4. Solution: A solution that contains 70 g of $\ce{KI}$ at $0^\circ\mathrm{C}$
- Solubility of $\ce{KI}$ at $0^\circ\mathrm{C}$: ~128 g/100 mL water.
- Since $70\ \text{g} < 128\ \text{g}$, the solution is unsaturated.
- No additional solute limit reached (can dissolve more).
Filled Table (Summarized):
| Solution | Saturated or Unsaturated | If Saturated: How much more solute can dissolve? |
|---|---|---|
| $\ce{NH4Cl}$ (50 g, $50^\circ\mathrm{C}$) | Saturated | 0 g (already saturated) |
| $\ce{KClO3}$ (20 g, $50^\circ\mathrm{C}$) | Saturated | 0 g (excess undissolved; max is 18 g) |
| $\ce{KI}$ (70 g, $0^\circ\mathrm{C}$) | Unsaturated | None (unsaturated) |
(Note: Solubility values are approximate from standard solubility curves. Adjust if precise values differ.)