QUESTION IMAGE
Question
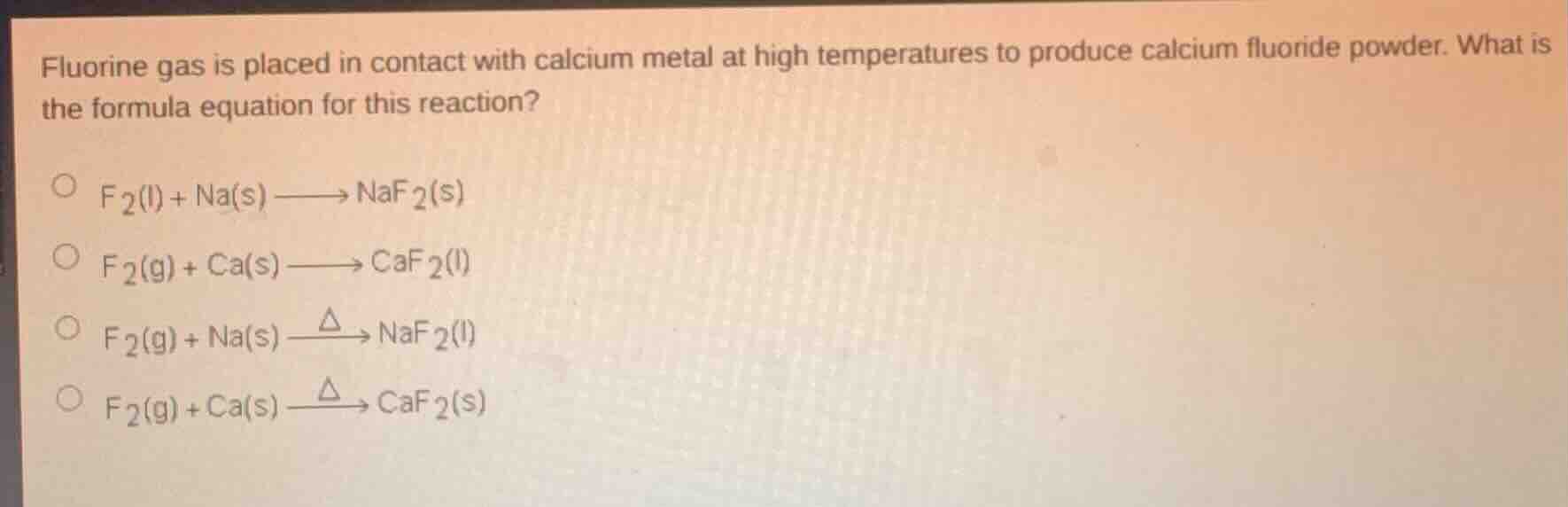
fluorine gas is placed in contact with calcium metal at high temperatures to produce calcium fluoride powder. what is the formula equation for this reaction?\
\\(\ce{f_{2}(l) + na(s) -> naf_{2}(s)}\\)\
\\(\ce{f_{2}(g) + ca(s) -> caf_{2}(l)}\\)\
\\(\ce{f_{2}(g) + na(s) ->\delta naf_{2}(l)}\\)\
\\(\ce{f_{2}(g) + ca(s) ->\delta caf_{2}(s)}\\)
- First, identify the reactants: Fluorine gas ($\ce{F2(g)}$) and calcium metal ($\ce{Ca(s)}$). The reaction occurs at high temperatures, so the $\Delta$ symbol (heat) should be included.
- The product is calcium fluoride powder, so its state is solid ($\ce{(s)}$). The formula for calcium fluoride is $\ce{CaF2}$ (calcium has a +2 charge, fluoride has a -1 charge, so 1 Ca and 2 F to balance charges).
- Now check the options:
- First option: Reactant is Na (sodium), not Ca, and product formula is wrong.
- Second option: No heat symbol, and product state is liquid (should be solid), and formula for calcium fluoride is correct but state and heat are missing.
- Third option: Reactant is Na, wrong metal, and product formula wrong.
- Fourth option: Reactants are $\ce{F2(g)}$ and $\ce{Ca(s)}$, heat symbol ($\Delta$) for high temperature, product is $\ce{CaF2(s)}$ (solid powder), which matches all criteria.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\ce{F2(g) + Ca(s) \stackrel{\Delta}{
ightarrow} CaF2(s)}$ (Note: In the original options, the fourth option is the correct one with the identifiers as per the problem's option numbering, assuming the last option is D here. If the options were labeled A - D, then D is the correct choice with the reaction equation $\ce{F2(g) + Ca(s) \stackrel{\Delta}{
ightarrow} CaF2(s)}$)