QUESTION IMAGE
Question
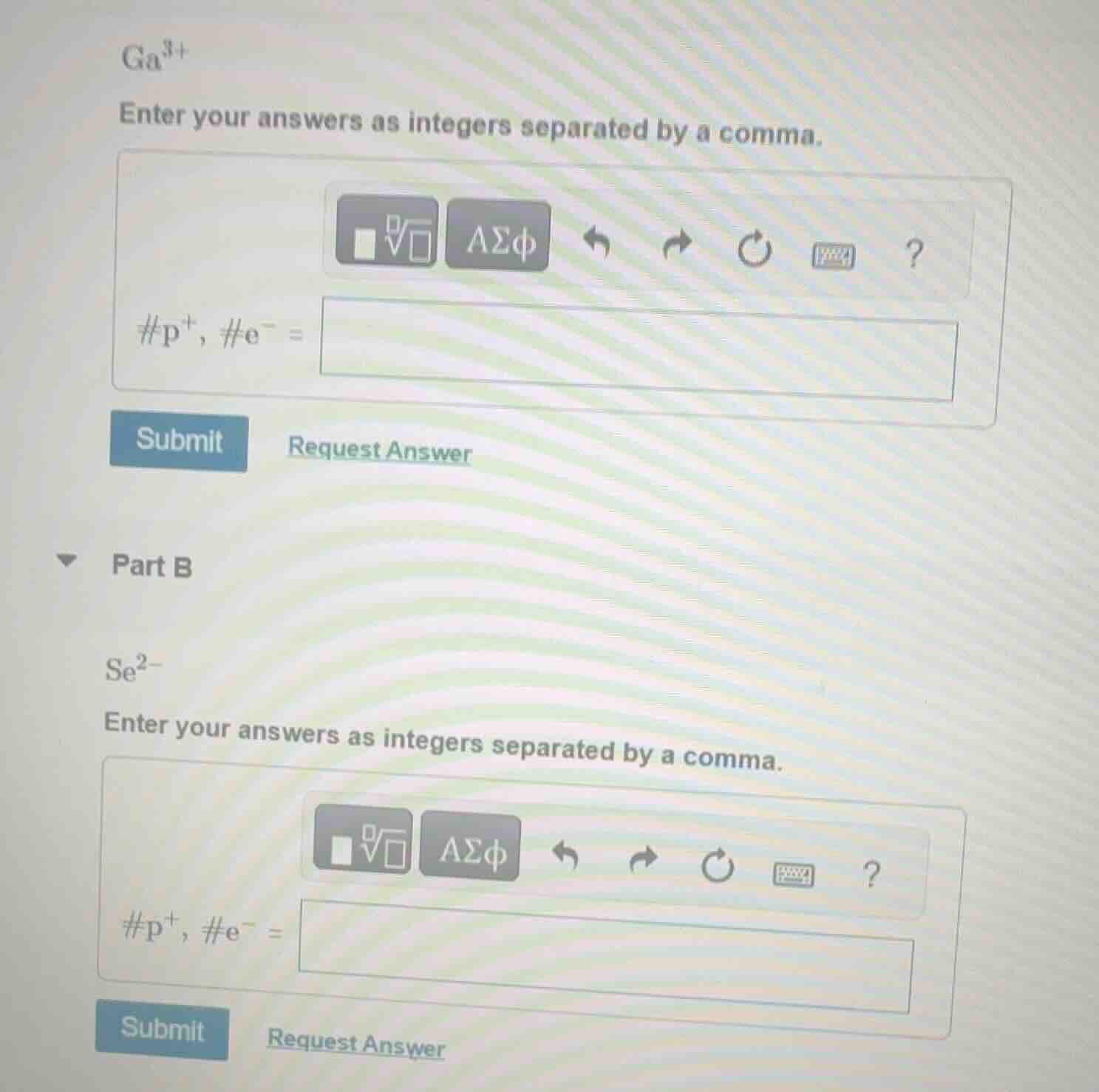
ga³⁺
enter your answers as integers separated by a comma.
#p⁺, #e⁻ =
submit request answer
part b
se²⁻
enter your answers as integers separated by a comma.
#p⁺, #e⁻ =
submit request answer
Step1: Find protons for Ga³⁺
Gallium (Ga) has an atomic number of 31, so $\#p^+ = 31$.
Step2: Find electrons for Ga³⁺
A +3 charge means 3 electrons lost: $\#e^- = 31 - 3 = 28$.
Step3: Find protons for Se²⁻
Selenium (Se) has an atomic number of 34, so $\#p^+ = 34$.
Step4: Find electrons for Se²⁻
A -2 charge means 2 electrons gained: $\#e^- = 34 + 2 = 36$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For $\text{Ga}^{3+}$: 31, 28
For $\text{Se}^{2-}$: 34, 36