QUESTION IMAGE
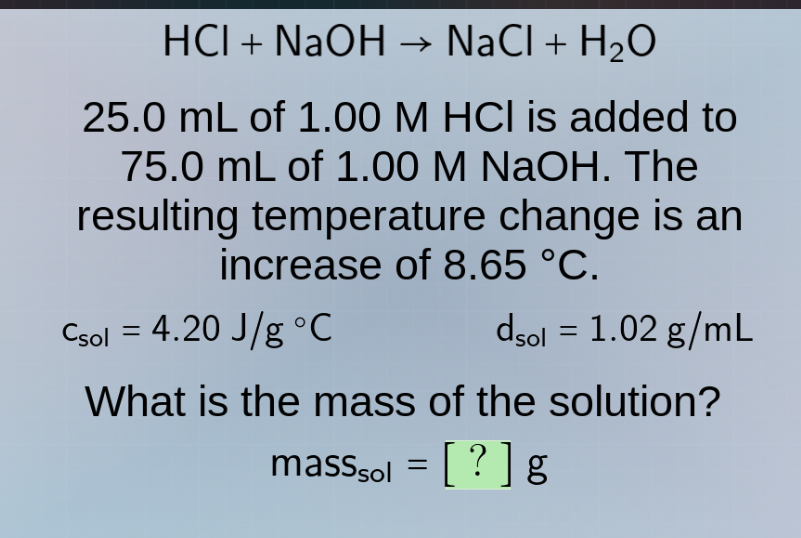
Question
hcl + naoh → nacl + h₂o
25.0 ml of 1.00 m hcl is added to
75.0 ml of 1.00 m naoh. the
resulting temperature change is an
increase of 8.65 °c.
(c_{\text{sol}} = 4.20 , \text{j/g °c}) (quadquadquad) (d_{\text{sol}} = 1.02 , \text{g/ml})
what is the mass of the solution?
( \text{mass}_{\text{sol}} = ? , \text{g} )
Step1: Find total volume of solution
The volume of HCl is 25.0 mL and NaOH is 75.0 mL. Total volume \( V = 25.0\space mL + 75.0\space mL = 100.0\space mL \).
Step2: Calculate mass using density formula
Density formula is \( d=\frac{m}{V} \), so \( m = d\times V \). Given \( d_{sol}=1.02\space g/mL \) and \( V = 100.0\space mL \), then \( m = 1.02\space g/mL\times100.0\space mL \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
102