QUESTION IMAGE
Question
how fda regulates artificial intelligence in medical products
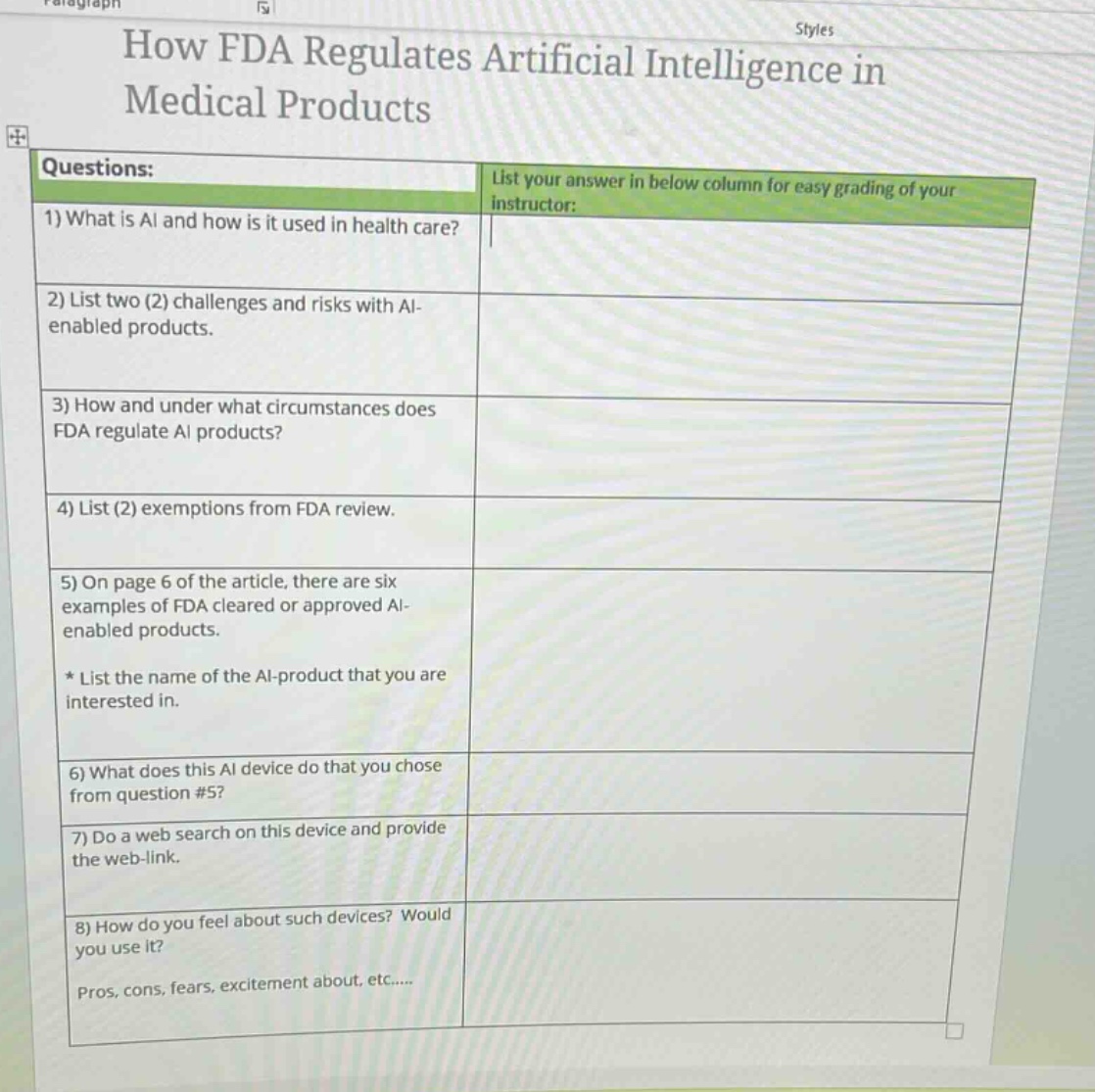
questions:
- what is ai and how is it used in health care?
- list two (2) challenges and risks with ai - enabled products.
- how and under what circumstances does fda regulate ai products?
- list (2) exemptions from fda review.
- on page 6 of the article, there are six examples of fda cleared or approved ai - enabled products.
- list the name of the ai - product that you are interested in.
- what does this ai device do that you chose from question #5?
- do a web search on this device and provide the web - link.
- how do you feel about such devices? would you use it?
pros, cons, fears, excitement about, etc.....
To answer these questions, we'll analyze each one based on knowledge of FDA regulation and AI in healthcare:
1) What is AI and how is it used in health care?
- AI (Artificial Intelligence): Systems or machines that mimic human intelligence to perform tasks (e.g., learning, problem - solving).
- Healthcare Use:
- Diagnosis: Analyze medical images (X - rays, MRIs) to detect diseases (e.g., cancer, fractures).
- Treatment Planning: Suggest personalized treatment plans based on patient data (genetics, medical history).
- Drug Discovery: Identify potential drug candidates and predict their efficacy.
- Monitoring: Track patient vitals (e.g., in wearable devices) to detect anomalies (like irregular heart rhythms).
2) List two (2) challenges and risks with AI - enabled products.
- Challenge 1: Bias in Data/Algorithms
AI systems learn from historical data. If this data has biases (e.g., underrepresentation of certain ethnic groups in medical datasets), the AI may produce biased results (e.g., misdiagnosing patients from underrepresented groups).
- Challenge 2: Lack of Transparency (Black Box Problem)
Many advanced AI models (e.g., deep learning) are “black boxes”—it’s hard to explain how they reach a decision. In healthcare, this makes it difficult for clinicians to trust or justify AI - driven recommendations to patients.
3) How and under what circumstances does FDA regulate AI products?
- Regulation Basis: The FDA regulates AI - enabled medical products as medical devices (under the Federal Food, Drug, and Cosmetic Act).
- Circumstances:
- If the AI product is intended for a medical purpose (e.g., diagnosis, treatment, disease prevention, or health monitoring), it falls under FDA oversight.
- The level of regulation (e.g., pre - market approval, 510(k) clearance) depends on the product’s risk classification:
- Low - risk: May get 510(k) clearance (proving it is as “substantially equivalent” to a legally marketed device).
- High - risk (e.g., AI for diagnosing life - threatening diseases): Requires pre - market approval (PMA), a more rigorous review of safety and effectiveness.
4) List (2) exemptions from FDA review.
- Exemption 1: Software as a Medical Device (SaMD) with Low Risk and No Significant Impact
Some low - risk SaMD (e.g., a simple app that only tracks daily steps and provides general health tips, with no medical diagnosis/treatment claims) may be exempt, especially if it meets the criteria of the FDA’s “Software Precertification Program” (for innovative, low - risk digital health tools).
- Exemption 2: Laboratory - Developed Tests (LDTs) in Some Cases
Historically, many LDTs (tests developed and used within a single laboratory) were exempt from FDA review. However, recent regulatory changes are narrowing this exemption, but some low - complexity LDTs may still fall outside full FDA review.
5) On page 6 of the article, there are six examples of FDA cleared or approved AI - enabled products. List the name of the AI - product that you are interested in.
(Note: Since the article’s page 6 is not provided, we’ll use a common example.)
Example: IBM Watson for Oncology (FDA - related for cancer diagnosis/treatment recommendations) or IDx - DR (an AI - powered device to detect diabetic retinopathy, FDA - approved). Let's pick IDx - DR for this example.
6) What does this AI device (IDx - DR) do that you chose from question #5?
IDx - DR is a diagnostic device that uses AI to analyze retinal images (taken with a fundus…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To answer these questions, we'll analyze each one based on knowledge of FDA regulation and AI in healthcare:
1) What is AI and how is it used in health care?
- AI (Artificial Intelligence): Systems or machines that mimic human intelligence to perform tasks (e.g., learning, problem - solving).
- Healthcare Use:
- Diagnosis: Analyze medical images (X - rays, MRIs) to detect diseases (e.g., cancer, fractures).
- Treatment Planning: Suggest personalized treatment plans based on patient data (genetics, medical history).
- Drug Discovery: Identify potential drug candidates and predict their efficacy.
- Monitoring: Track patient vitals (e.g., in wearable devices) to detect anomalies (like irregular heart rhythms).
2) List two (2) challenges and risks with AI - enabled products.
- Challenge 1: Bias in Data/Algorithms
AI systems learn from historical data. If this data has biases (e.g., underrepresentation of certain ethnic groups in medical datasets), the AI may produce biased results (e.g., misdiagnosing patients from underrepresented groups).
- Challenge 2: Lack of Transparency (Black Box Problem)
Many advanced AI models (e.g., deep learning) are “black boxes”—it’s hard to explain how they reach a decision. In healthcare, this makes it difficult for clinicians to trust or justify AI - driven recommendations to patients.
3) How and under what circumstances does FDA regulate AI products?
- Regulation Basis: The FDA regulates AI - enabled medical products as medical devices (under the Federal Food, Drug, and Cosmetic Act).
- Circumstances:
- If the AI product is intended for a medical purpose (e.g., diagnosis, treatment, disease prevention, or health monitoring), it falls under FDA oversight.
- The level of regulation (e.g., pre - market approval, 510(k) clearance) depends on the product’s risk classification:
- Low - risk: May get 510(k) clearance (proving it is as “substantially equivalent” to a legally marketed device).
- High - risk (e.g., AI for diagnosing life - threatening diseases): Requires pre - market approval (PMA), a more rigorous review of safety and effectiveness.
4) List (2) exemptions from FDA review.
- Exemption 1: Software as a Medical Device (SaMD) with Low Risk and No Significant Impact
Some low - risk SaMD (e.g., a simple app that only tracks daily steps and provides general health tips, with no medical diagnosis/treatment claims) may be exempt, especially if it meets the criteria of the FDA’s “Software Precertification Program” (for innovative, low - risk digital health tools).
- Exemption 2: Laboratory - Developed Tests (LDTs) in Some Cases
Historically, many LDTs (tests developed and used within a single laboratory) were exempt from FDA review. However, recent regulatory changes are narrowing this exemption, but some low - complexity LDTs may still fall outside full FDA review.
5) On page 6 of the article, there are six examples of FDA cleared or approved AI - enabled products. List the name of the AI - product that you are interested in.
(Note: Since the article’s page 6 is not provided, we’ll use a common example.)
Example: IBM Watson for Oncology (FDA - related for cancer diagnosis/treatment recommendations) or IDx - DR (an AI - powered device to detect diabetic retinopathy, FDA - approved). Let's pick IDx - DR for this example.
6) What does this AI device (IDx - DR) do that you chose from question #5?
IDx - DR is a diagnostic device that uses AI to analyze retinal images (taken with a fundus camera). It automatically detects signs of diabetic retinopathy, a complication of diabetes that can lead to blindness. It provides a “positive” or “negative” result for the presence of the disease, helping eye care providers (even those without specialized retinopathy expertise) to screen patients.
7) Do a web search on this device and provide the web - link.
A reliable web - link for IDx - DR is from the FDA’s official site or the company’s site:
https://www.fda.gov/news - events/press - announcements/fda - approves - first - ai - device - detect - diabetic - retinopathy
8) How do you feel about such devices? Would you use it? Pros, cons, fears, excitement about, etc.....
- Pros:
- Accessibility: Devices like IDx - DR expand access to specialized care (e.g., in rural areas with few eye specialists).
- Accuracy: AI can detect subtle signs of disease that human eyes might miss, improving early diagnosis.
- Cons/Fears:
- Over - reliance: Clinicians might rely too heavily on AI and neglect their own clinical judgment.
- Data Privacy: AI devices handle sensitive patient data (retinal images, medical history), raising concerns about data breaches or misuse.
- Would I Use It?
Yes, I would use it (or support its use in healthcare). The benefits (early detection, increased access) outweigh the risks, especially with proper safeguards (e.g., data encryption, clinician oversight) in place. The excitement of using technology to save vision and improve health is significant, though I’d want to ensure it’s used as a tool, not a replacement for human care.
Final Answers (Summarized for Each Question):
- AI is a system/machine mimicking human intelligence; used in healthcare for diagnosis, treatment planning, drug discovery, and patient monitoring.
- Bias in data/algorithms; lack of transparency (black box problem).
- FDA regulates AI medical products as devices; based on medical purpose and risk classification (low - risk: 510(k); high - risk: PMA).
- Low - risk SaMD with no medical claims; some low - complexity LDTs (historical exemption, evolving).
- IDx - DR (example).
- IDx - DR analyzes retinal images to detect diabetic retinopathy.
- https://www.fda.gov/news - events/press - announcements/fda - approves - first - ai - device - detect - diabetic - retinopathy
- Positive overall; would use (or support use) due to accessibility and accuracy benefits, with concerns about over - reliance and data privacy.