QUESTION IMAGE
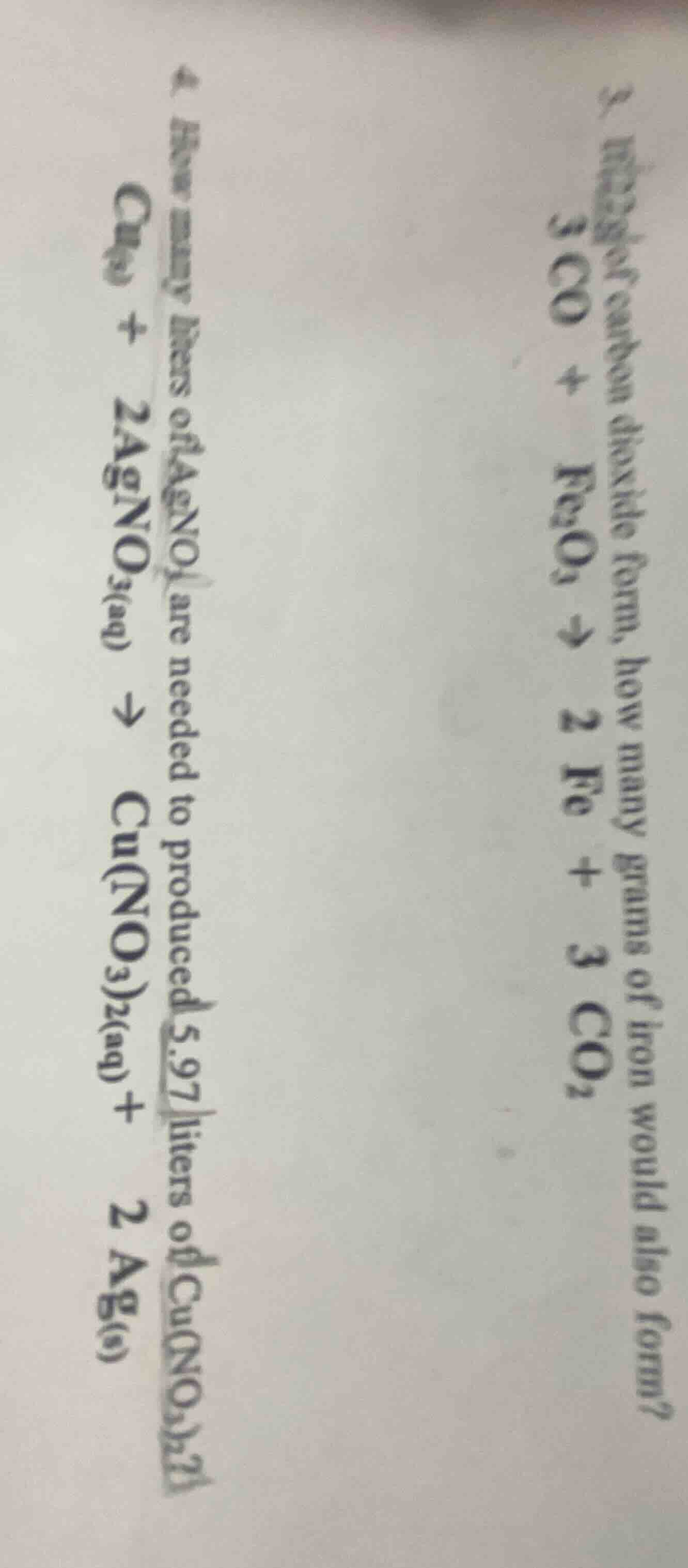
Question
- how many grams of carbon dioxide form, how many grams of iron would also form? \\(3co + fe_2o_3 \
ightarrow 2fe + 3co_2\\)\
- how many liters of \\(agno_3\\) are needed to produced 5.97 liters of \\(cu(no_3)_2\\)? \\(cu_{(s)} + 2agno_{3(aq)} \
ightarrow cu(no_3)_{2(aq)} + 2ag_{(s)}\\)
(Problem 3):
Step1: Moles of $\text{CO}_2$ calculation
Molar mass of $\text{CO}_2 = 12 + 2\times16 = 44\ \text{g/mol}$
Moles of $\text{CO}_2 = \frac{1022\ \text{g}}{44\ \text{g/mol}} = 23.227\ \text{mol}$
Step2: Mole ratio of $\text{Fe}$ to $\text{CO}_2$
From reaction: $\frac{n(\text{Fe})}{n(\text{CO}_2)} = \frac{2}{3}$
Step3: Moles of $\text{Fe}$ calculation
$n(\text{Fe}) = \frac{2}{3} \times n(\text{CO}_2) = \frac{2}{3} \times 23.227\ \text{mol} = 15.485\ \text{mol}$
Step4: Mass of $\text{Fe}$ calculation
Molar mass of $\text{Fe} = 55.85\ \text{g/mol}$
Mass of $\text{Fe} = 15.485\ \text{mol} \times 55.85\ \text{g/mol}$
(Problem 4):
Step1: Mole ratio of $\text{AgNO}_3$ to $\text{Cu(NO}_3\text{)}_2$
From reaction: $\frac{n(\text{AgNO}_3)}{n(\text{Cu(NO}_3\text{)}_2)} = \frac{2}{1}$
Step2: Volume of $\text{AgNO}_3$ calculation
$V(\text{AgNO}_3) = 2 \times V(\text{Cu(NO}_3\text{)}_2) = 2 \times 5.97\ \text{L}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Problem 3: $864.8\ \text{g}$
Problem 4: $11.94\ \text{L}$