QUESTION IMAGE
Question
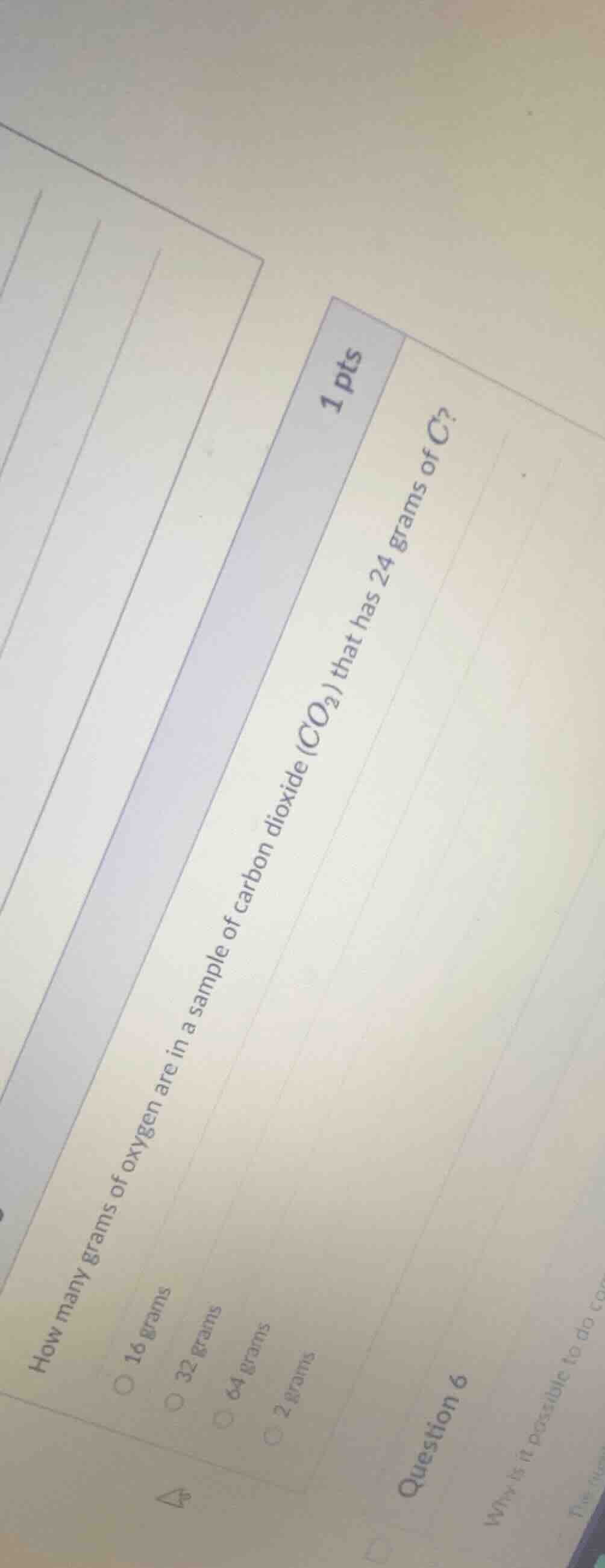
how many grams of oxygen are in a sample of carbon dioxide (co₂) that has 24 grams of c?
16 grams
32 grams
64 grams
2 grams
Step1: Find molar mass of $\text{CO}_2$
Molar mass of $\text{C} = 12\ \text{g/mol}$, molar mass of $\text{O} = 16\ \text{g/mol}$.
Molar mass of $\text{CO}_2 = 12 + 2\times16 = 44\ \text{g/mol}$
Step2: Find mass ratio of $\text{O}$ to $\text{C}$ in $\text{CO}_2$
Mass of $\text{O}$ per $\text{CO}_2$: $32\ \text{g}$, mass of $\text{C}$ per $\text{CO}_2$: $12\ \text{g}$.
Ratio $\frac{\text{mass of O}}{\text{mass of C}} = \frac{32}{12} = \frac{8}{3}$
Step3: Calculate mass of $\text{O}$
Given mass of $\text{C} = 24\ \text{g}$.
Mass of $\text{O} = 24 \times \frac{8}{3}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
64 grams