QUESTION IMAGE
Question
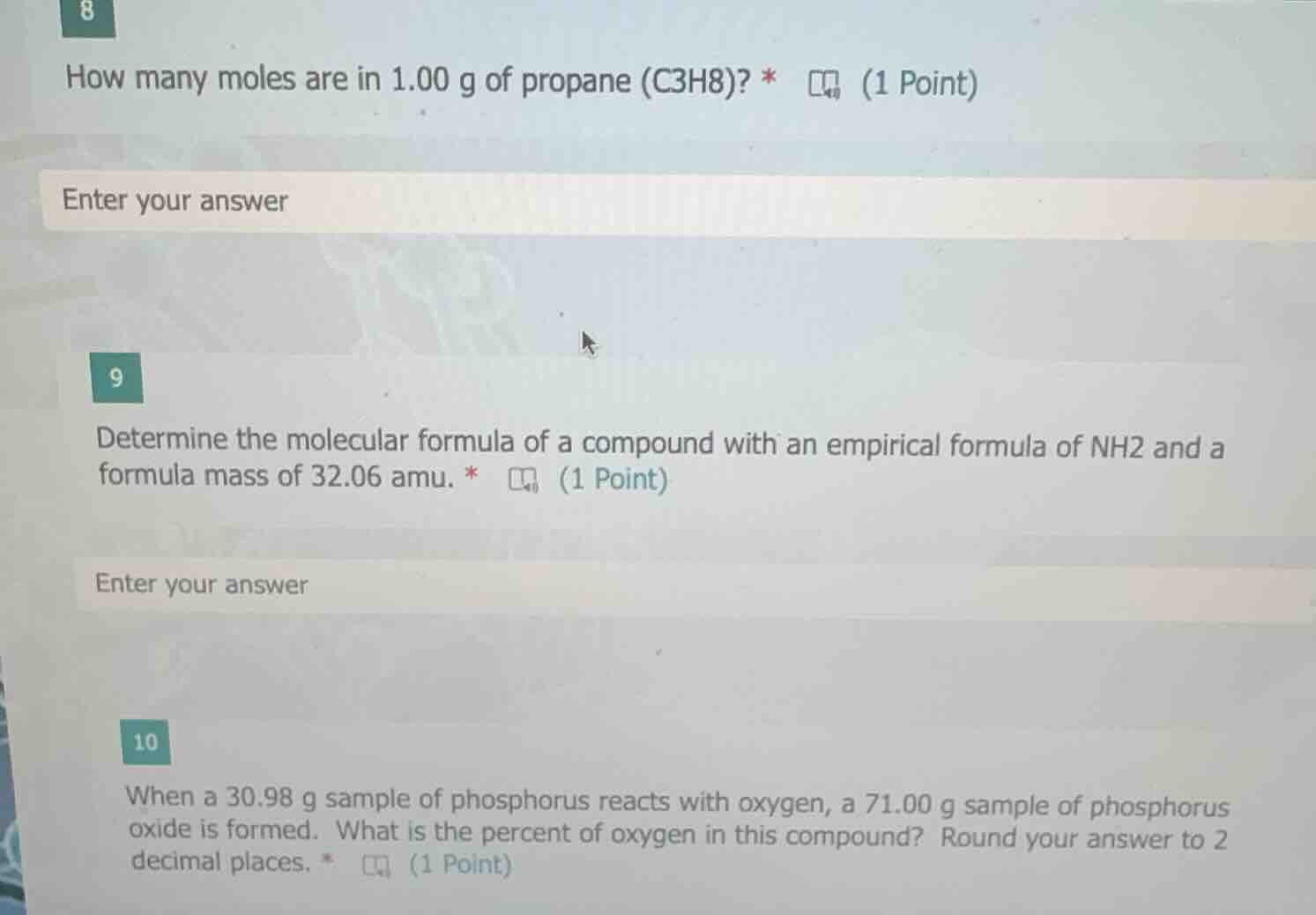
8
how many moles are in 1.00 g of propane (c3h8)? *
enter your answer
9
determine the molecular formula of a compound with an empirical formula of nh2 and a formula mass of 32.06 amu. *
enter your answer
10
when a 30.98 g sample of phosphorus reacts with oxygen, a 71.00 g sample of phosphorus oxide is formed. what is the percent of oxygen in this compound? round your answer to 2 decimal places. *
Question 8
Step1: Calculate molar mass of C₃H₈
Molar mass of C: \( 12.01 \, \text{g/mol} \), H: \( 1.008 \, \text{g/mol} \).
For \( \text{C}_3\text{H}_8 \), molar mass \( = 3\times12.01 + 8\times1.008 \)
\( = 36.03 + 8.064 = 44.094 \, \text{g/mol} \)
Step2: Use moles formula (\( n = \frac{m}{M} \))
Given \( m = 1.00 \, \text{g} \), \( M = 44.094 \, \text{g/mol} \)
\( n = \frac{1.00}{44.094} \approx 0.0227 \, \text{mol} \)
Step1: Calculate empirical formula mass of NH₂
Molar mass of N: \( 14.01 \, \text{g/mol} \), H: \( 1.008 \, \text{g/mol} \)
Empirical mass \( = 14.01 + 2\times1.008 = 14.01 + 2.016 = 16.026 \, \text{amu} \)
Step2: Find ratio (\( n = \frac{\text{Molecular mass}}{\text{Empirical mass}} \))
Given molecular mass \( = 32.06 \, \text{amu} \)
\( n = \frac{32.06}{16.026} \approx 2 \)
Step3: Multiply empirical formula by \( n \)
Molecular formula: \( (\text{NH}_2)_2 = \text{N}_2\text{H}_4 \)
Step1: Find mass of oxygen
Mass of compound (phosphorus oxide) \( = 71.00 \, \text{g} \)
Mass of phosphorus \( = 30.98 \, \text{g} \)
Mass of oxygen \( = 71.00 - 30.98 = 40.02 \, \text{g} \)
Step2: Calculate percent oxygen (\( \% \text{O} = \frac{\text{mass O}}{\text{mass compound}} \times 100 \))
\( \% \text{O} = \frac{40.02}{71.00} \times 100 \approx 56.37\% \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 0.0227 \) (or rounded as needed, e.g., 0.0227 mol)