QUESTION IMAGE
Question
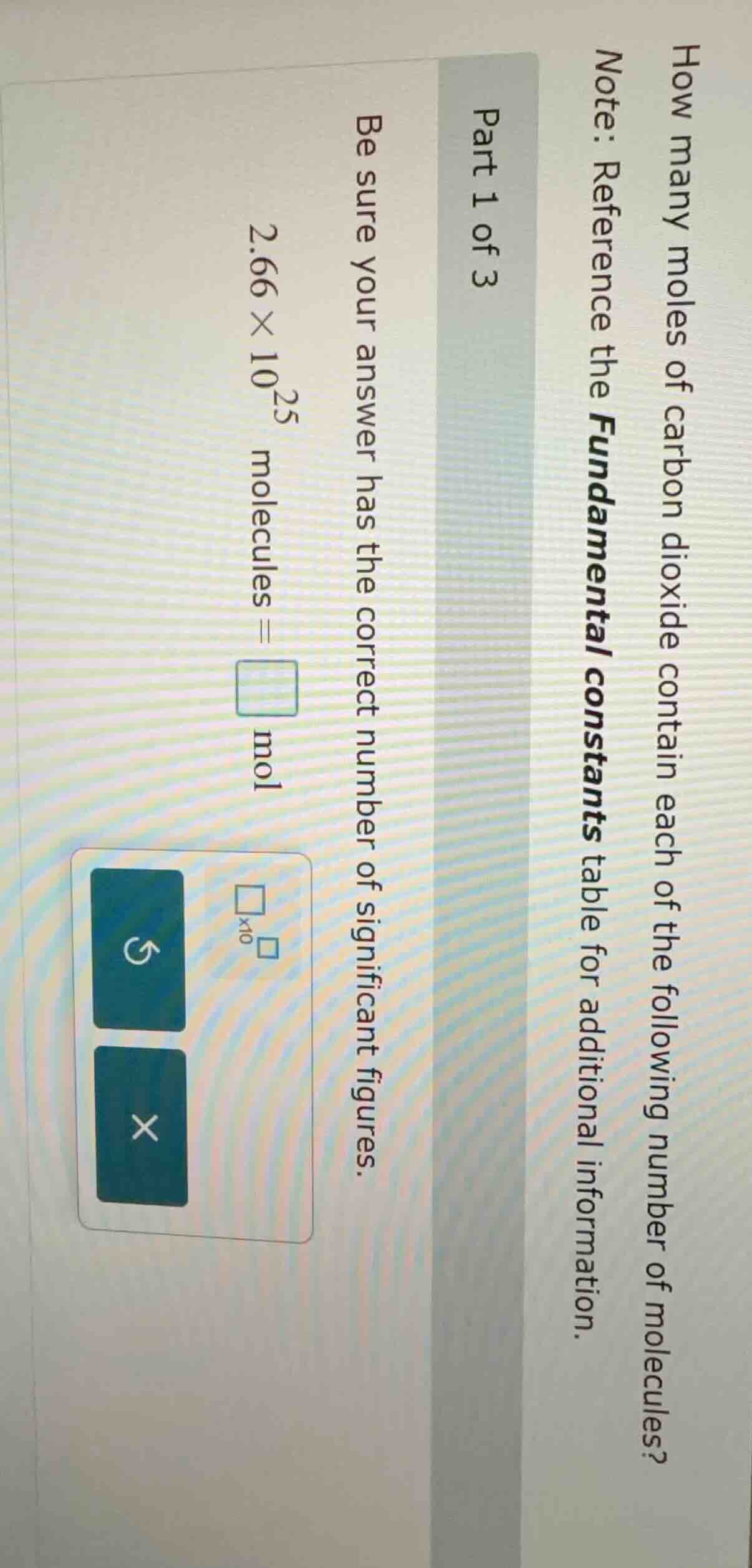
how many moles of carbon dioxide contain each of the following number of molecules? note: reference the fundamental constants table for additional information. part 1 of 3 be sure your answer has the correct number of significant figures. \\(2.66 \times 10^{25}\\) molecules = \\(\square\\) mol
Step1: Recall Avogadro's number
Avogadro's number is \( 6.022 \times 10^{23} \) molecules/mol. To find the number of moles, we divide the number of molecules by Avogadro's number.
\[
\text{Moles} = \frac{\text{Number of molecules}}{\text{Avogadro's number}}
\]
Step2: Substitute the values
Given the number of molecules is \( 2.66 \times 10^{25} \) and Avogadro's number is \( 6.022 \times 10^{23} \) molecules/mol.
\[
\text{Moles} = \frac{2.66 \times 10^{25}}{6.022 \times 10^{23}}
\]
Step3: Perform the division
First, divide the coefficients: \( \frac{2.66}{6.022} \approx 0.4417 \). Then, divide the powers of 10: \( 10^{25 - 23} = 10^{2} \). Multiply these results together: \( 0.4417 \times 10^{2} = 44.17 \) (rounded to the correct number of significant figures, the given value \( 2.66 \times 10^{25} \) has 3 significant figures, so the answer should have 3 significant figures).
\[
\text{Moles} \approx 44.2
\]
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 44.2 \)