QUESTION IMAGE
Question
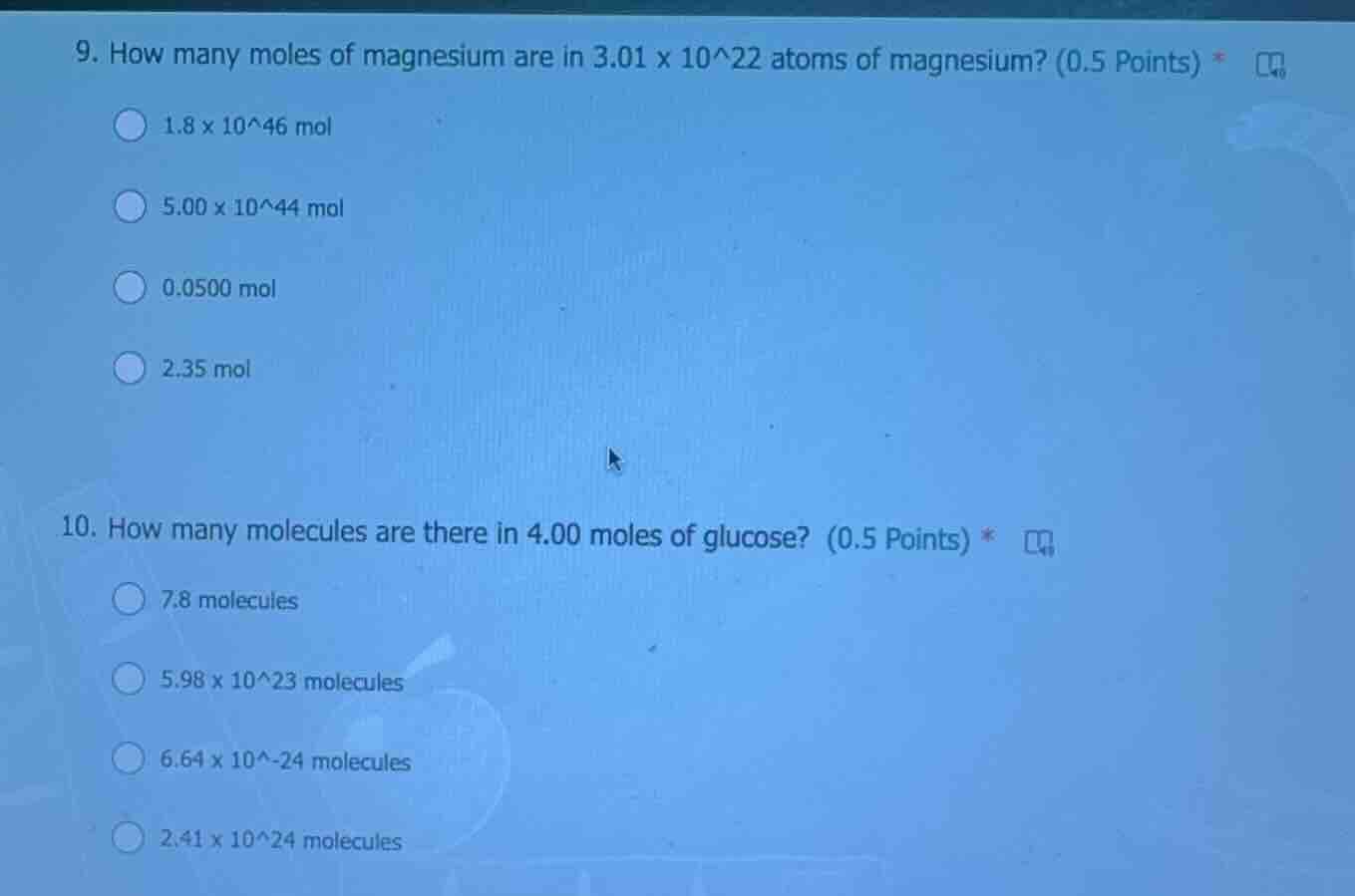
- how many moles of magnesium are in 3.01 x 10^22 atoms of magnesium? (0.5 points) *
1.8 x 10^46 mol
5.00 x 10^44 mol
0.0500 mol
2.35 mol
- how many molecules are there in 4.00 moles of glucose? (0.5 points) *
7.8 molecules
5.98 x 10^23 molecules
6.64 x 10^-24 molecules
2.41 x 10^24 molecules
Question 9
Step1: Recall Avogadro's number
Avogadro's number is \( 6.02 \times 10^{23} \) atoms/mol. To find moles from atoms, use the formula: \( \text{Moles} = \frac{\text{Number of atoms}}{\text{Avogadro's number}} \)
Step2: Substitute values
Given number of atoms \( = 3.01 \times 10^{22} \), Avogadro's number \( = 6.02 \times 10^{23} \) atoms/mol.
\( \text{Moles} = \frac{3.01 \times 10^{22}}{6.02 \times 10^{23}} \)
Step3: Calculate the result
Simplify the fraction: \( \frac{3.01}{6.02} = 0.5 \), and \( \frac{10^{22}}{10^{23}} = 10^{-1} = 0.1 \). So \( 0.5 \times 0.1 = 0.0500 \) mol.
Step1: Recall Avogadro's number
Avogadro's number is \( 6.02 \times 10^{23} \) molecules/mol. To find molecules from moles, use the formula: \( \text{Number of molecules} = \text{Moles} \times \text{Avogadro's number} \)
Step2: Substitute values
Given moles of glucose \( = 4.00 \) mol, Avogadro's number \( = 6.02 \times 10^{23} \) molecules/mol.
\( \text{Number of molecules} = 4.00 \times 6.02 \times 10^{23} \)
Step3: Calculate the result
\( 4.00 \times 6.02 = 24.08 \), so \( 24.08 \times 10^{23} = 2.408 \times 10^{24} \approx 2.41 \times 10^{24} \) molecules.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.0500 mol