QUESTION IMAGE
Question
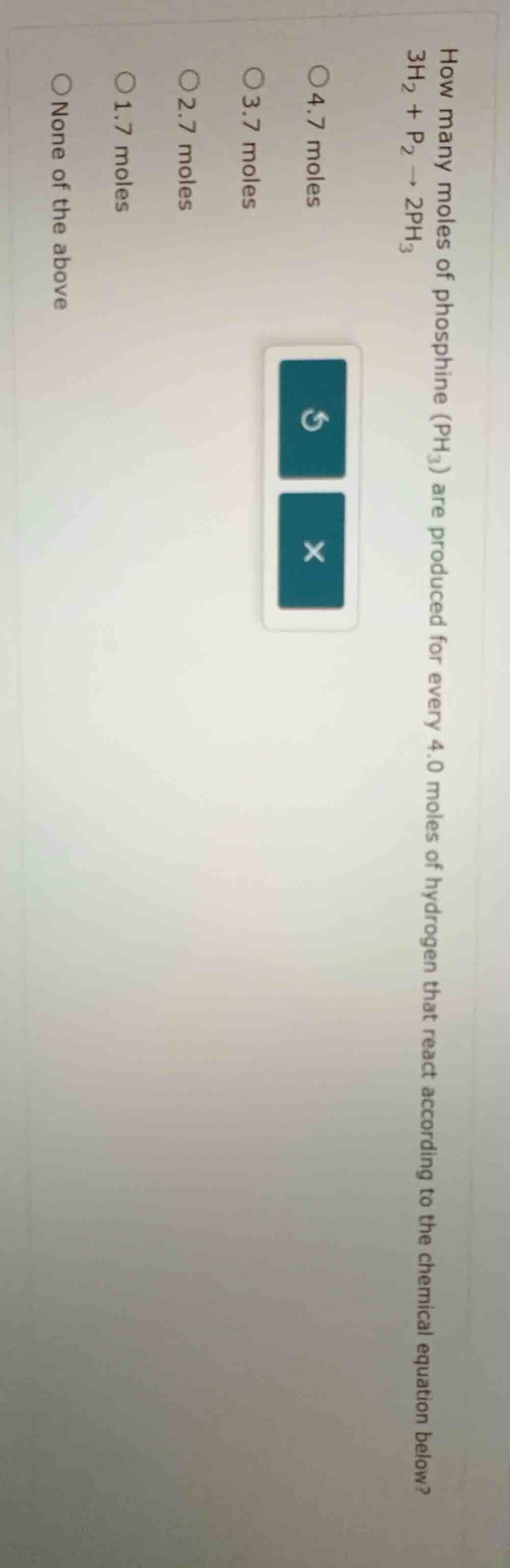
how many moles of phosphine (ph₃) are produced for every 4.0 moles of hydrogen that react according to the chemical equation below? 3h₂ + p₂ → 2ph₃ 4.7 moles 3.7 moles 2.7 moles 1.7 moles none of the above
Step1: Identify mole ratio
From the equation \( 3H_2 + P_2
ightarrow 2PH_3 \), the mole ratio of \( H_2 \) to \( PH_3 \) is \( 3:2 \).
Step2: Set up proportion
Let \( x \) be moles of \( PH_3 \). Proportion: \( \frac{3}{2}=\frac{4.0}{x} \) (wait, no—correct ratio: \( 3 \) moles \( H_2 \) produce \( 2 \) moles \( PH_3 \). So \( \frac{\text{moles of } H_2}{\text{moles of } PH_3}=\frac{3}{2} \). Given moles of \( H_2 = 4.0 \), solve for \( x \): \( x=\frac{2\times4.0}{3} \)
Step3: Calculate
\( x=\frac{8.0}{3}\approx2.7 \) moles.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2.7 moles (Option: 2.7 moles)