QUESTION IMAGE
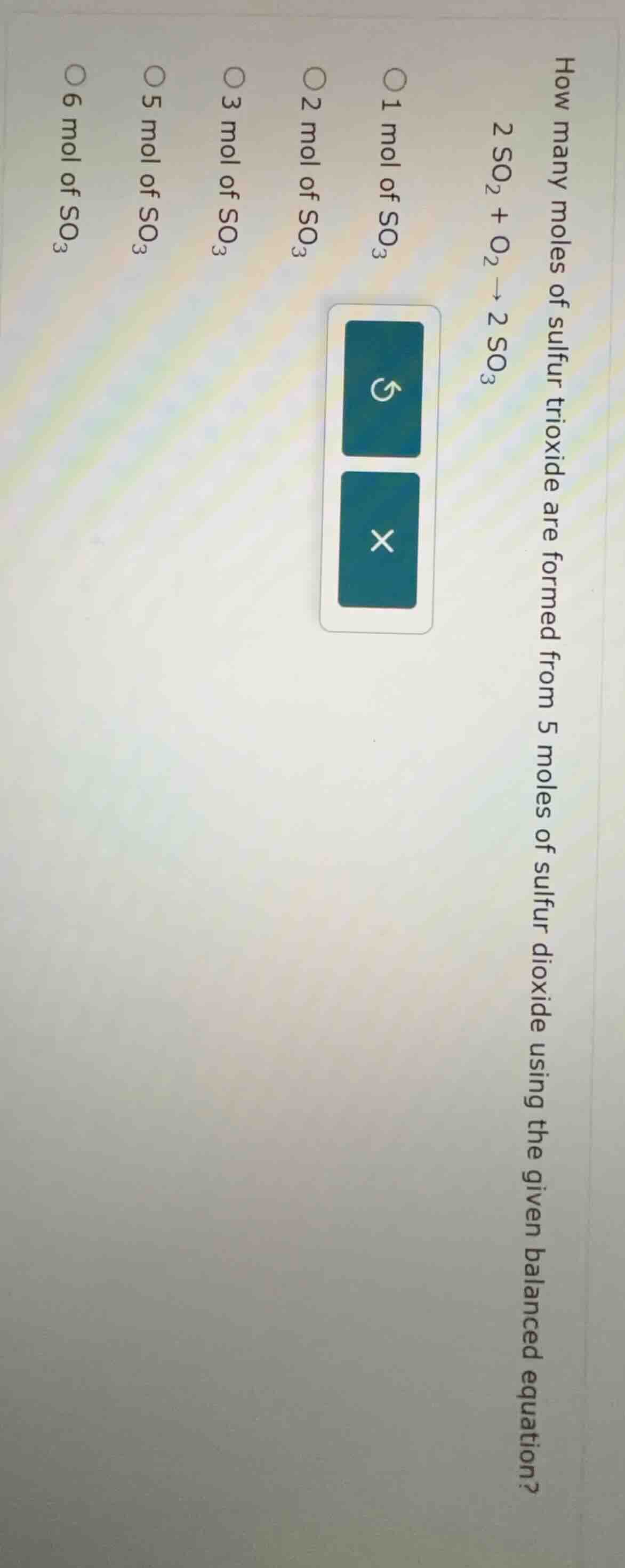
Question
how many moles of sulfur trioxide are formed from 5 moles of sulfur dioxide using the given balanced equation?
2 so₂ + o₂ → 2 so₃
1 mol of so₃
2 mol of so₃
3 mol of so₃
5 mol of so₃
6 mol of so₃
Step1: Analyze the balanced equation
The balanced chemical equation is \(2\text{SO}_2 + \text{O}_2
ightarrow 2\text{SO}_3\). From this equation, we can see that the mole ratio of \(\text{SO}_2\) to \(\text{SO}_3\) is \(2:2\) (or simplified to \(1:1\)).
Step2: Use the mole ratio to calculate moles of \(\text{SO}_3\)
We are given 5 moles of \(\text{SO}_2\). Let \(x\) be the moles of \(\text{SO}_3\) formed. Using the mole ratio \(\frac{\text{moles of } \text{SO}_2}{\text{moles of } \text{SO}_3}=\frac{2}{2} = 1\), we can set up the proportion \(\frac{5}{x}=\frac{2}{2}\). Cross - multiplying gives \(2x = 5\times2\), so \(x = 5\) moles.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
5 mol of \(SO_3\) (the option: 5 mol of \(SO_3\))